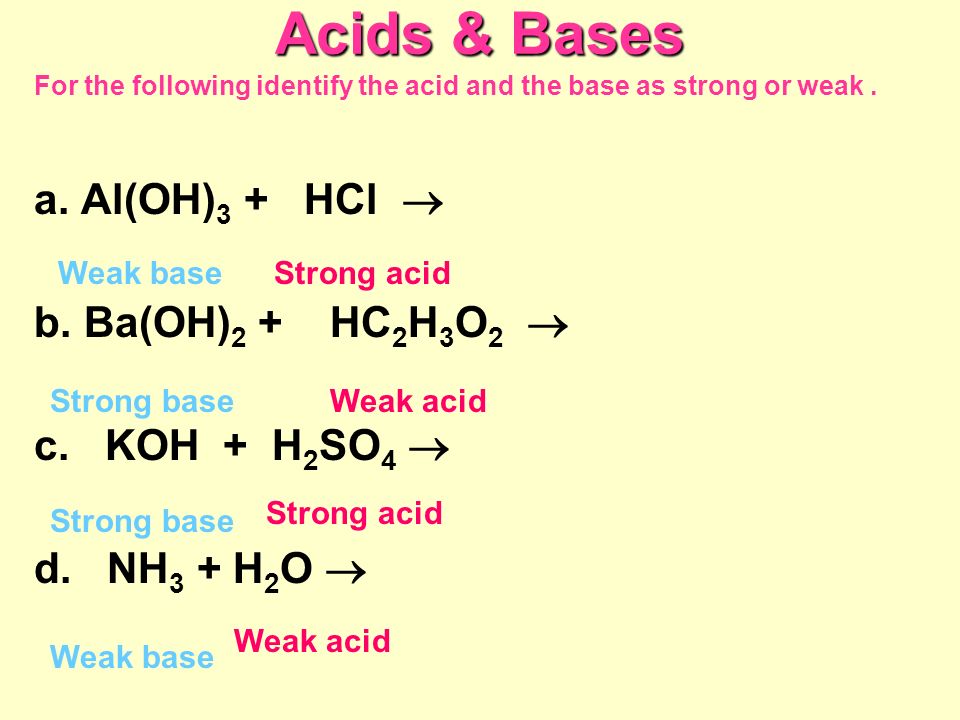
SOLVED: Consider the acid-base reaction 2 HNOz (aq) + Ba(OH)2 ( (aq) 4 Predict the products of the acid base reaction. 0 Ba? 2+ (aq) + NO; (aq) 0 H,BaNOs(aq) 0 Ba(NOzh(aq) +

E790: Acid/Base – Conductimetric Titration – Ba(OH)2 + H2SO4 | Lecture Demonstration Manual General Chemistry | University of Colorado Boulder

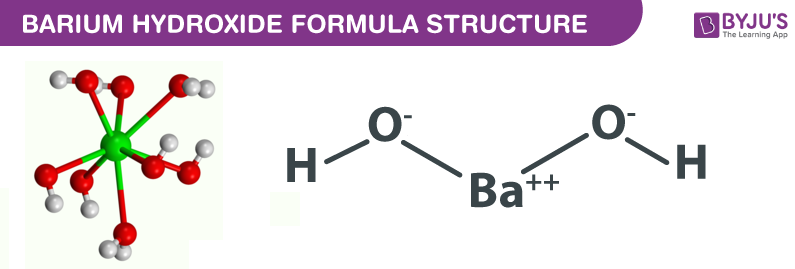
Calculate the pH of a 0.10 M solution of barium hydroxide, Ba(OH)2. Express your answer numerically - Home Work Help - Learn CBSE Forum

A. Calculate the pH of a 0.10 M solution of barium hydroxide, Ba(OH)2 - Home Work Help - Learn CBSE Forum

SOLVED: A solution of Ba(OH)2 was standardized against 0.1215 g of benzoic acid with grade of primary standard, C6H5COOH (122.12 g / mol). The end point was observed after adding 43.25 mL

A. Calculate the pH of a 0.10 M solution of barium hydroxide, Ba(OH)2 - Home Work Help - Learn CBSE Forum