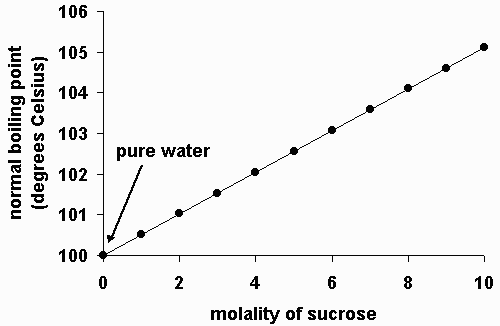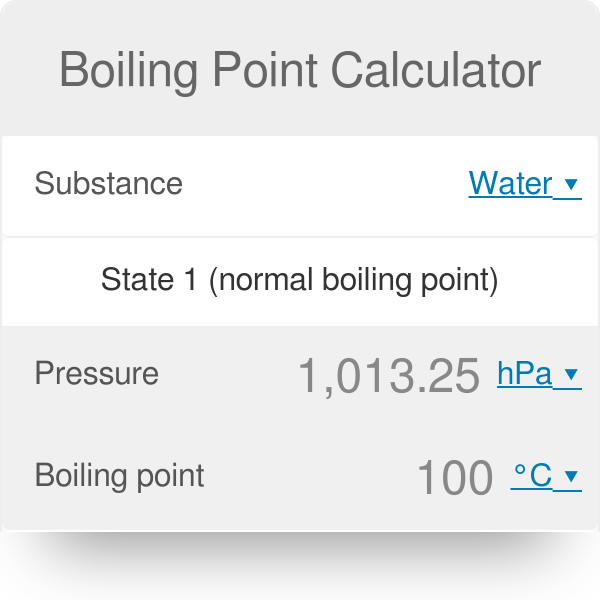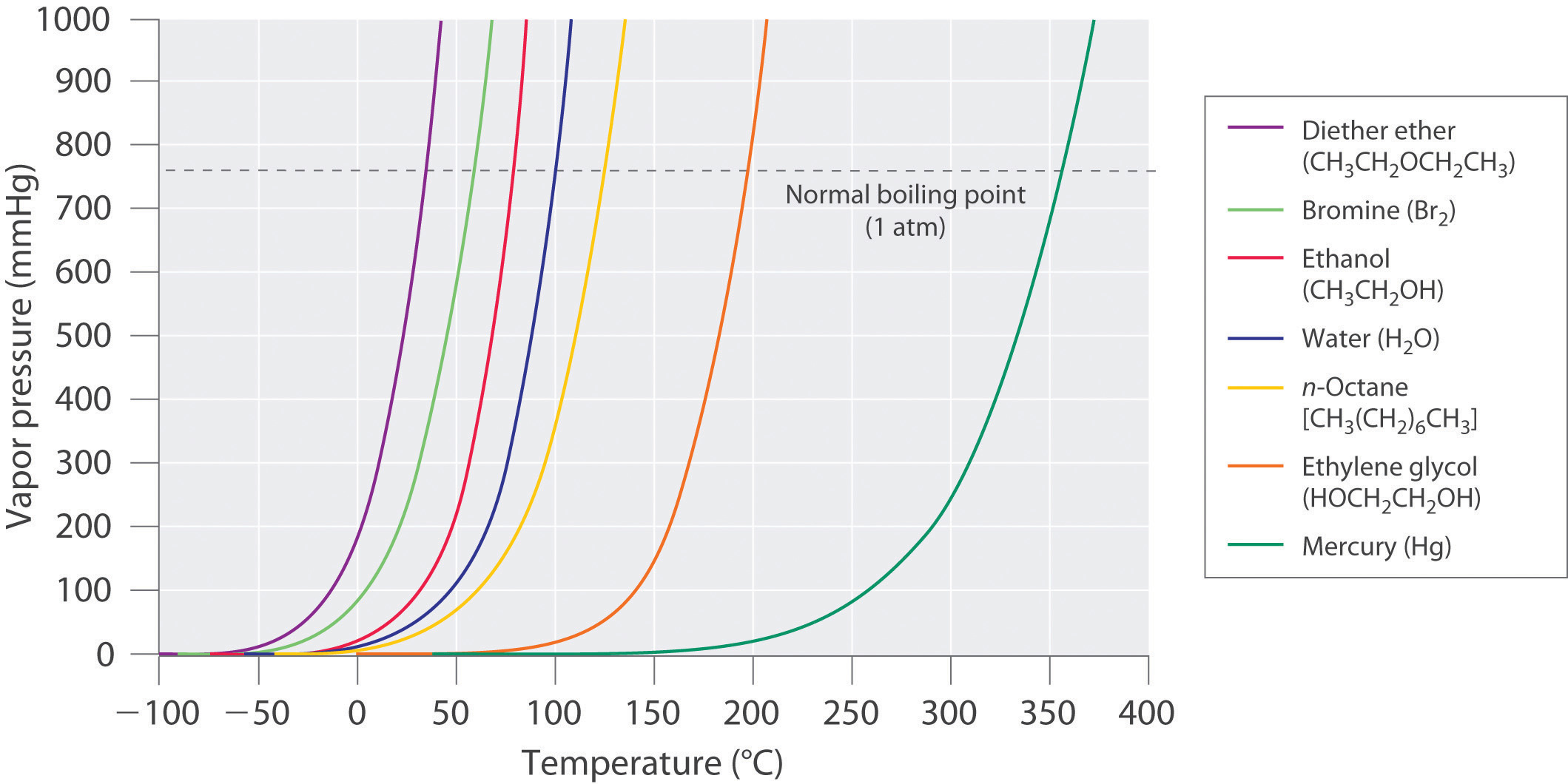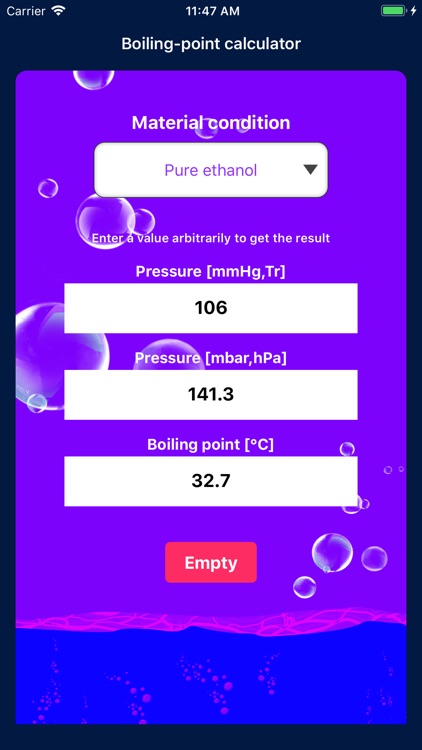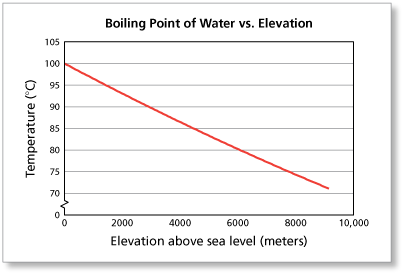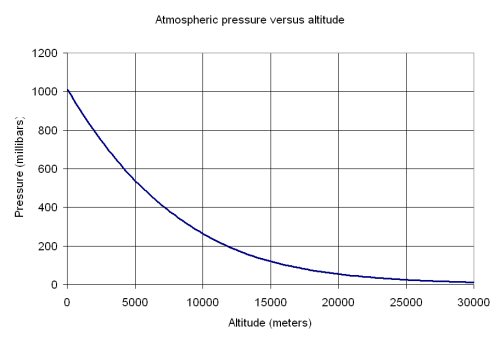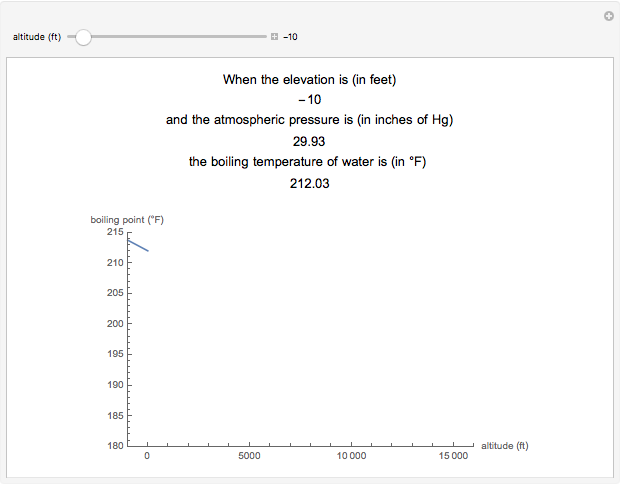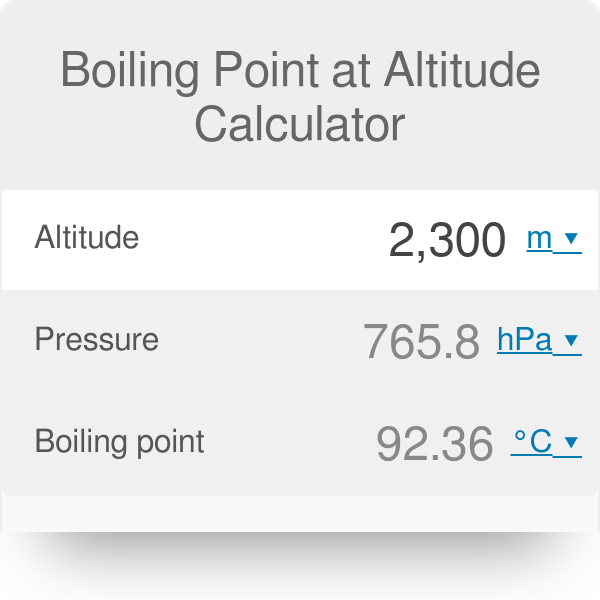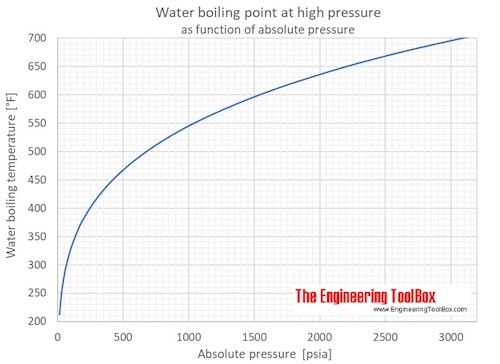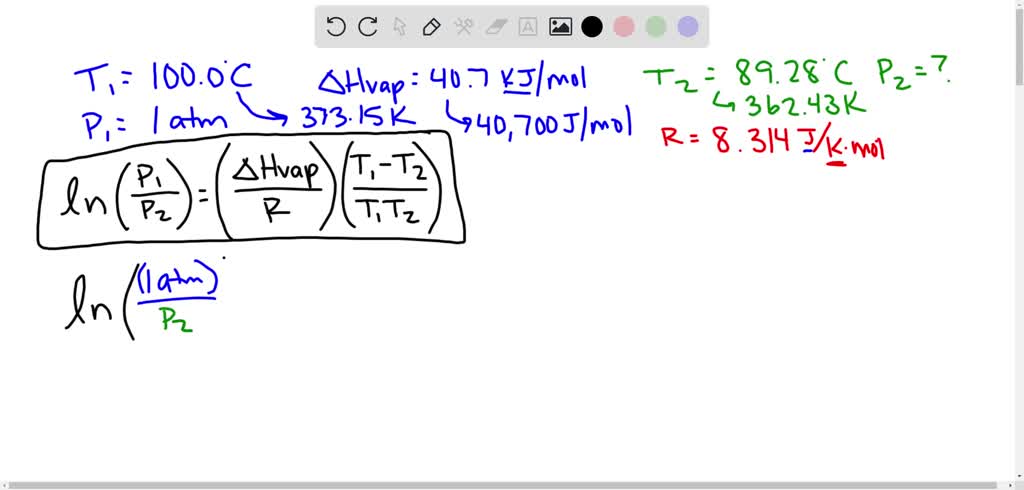
SOLVED: Suppose the boiling point of pure water at high altitude is 89.28 °C. Use the Clausius–Clapeyron equation to determine the atmospheric pressure at this high altitude. The normal boiling point of

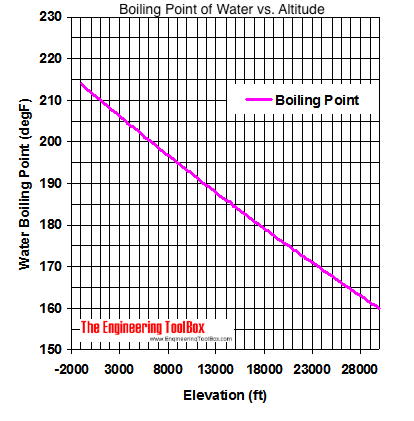
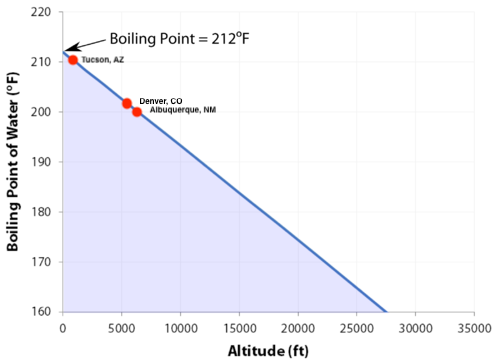
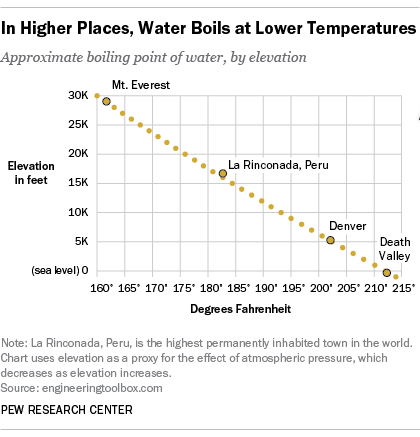
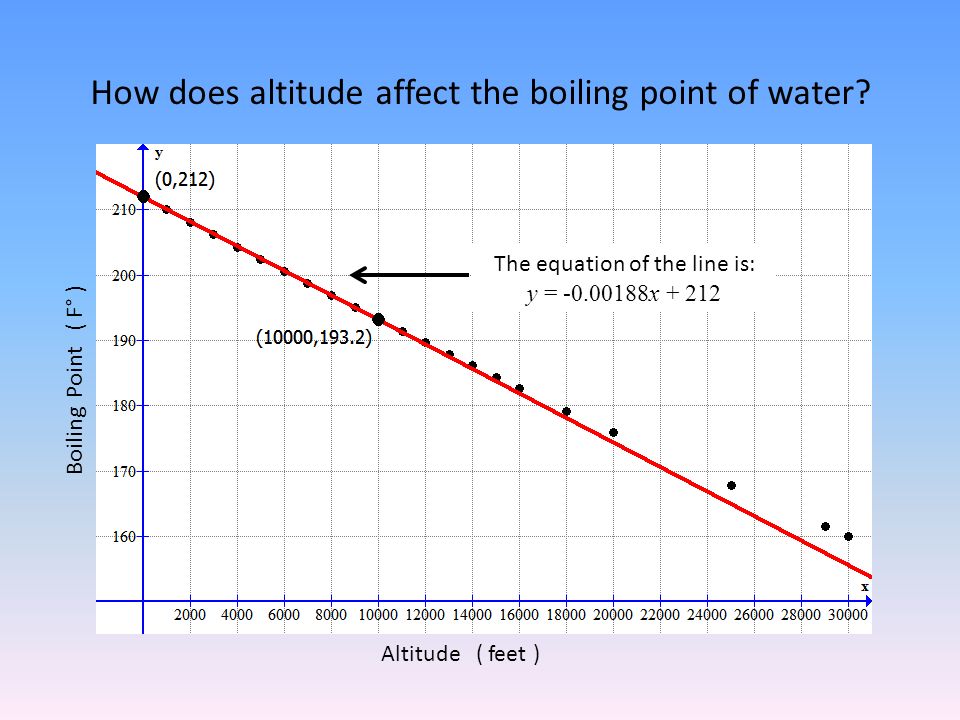
SOLVED: Boiling Point The relationship between altitude and the boiling point of water is linear. At an altitude of 8000 ft, water boils at 197.6^∘F. At an altitude of 4500 ft, water