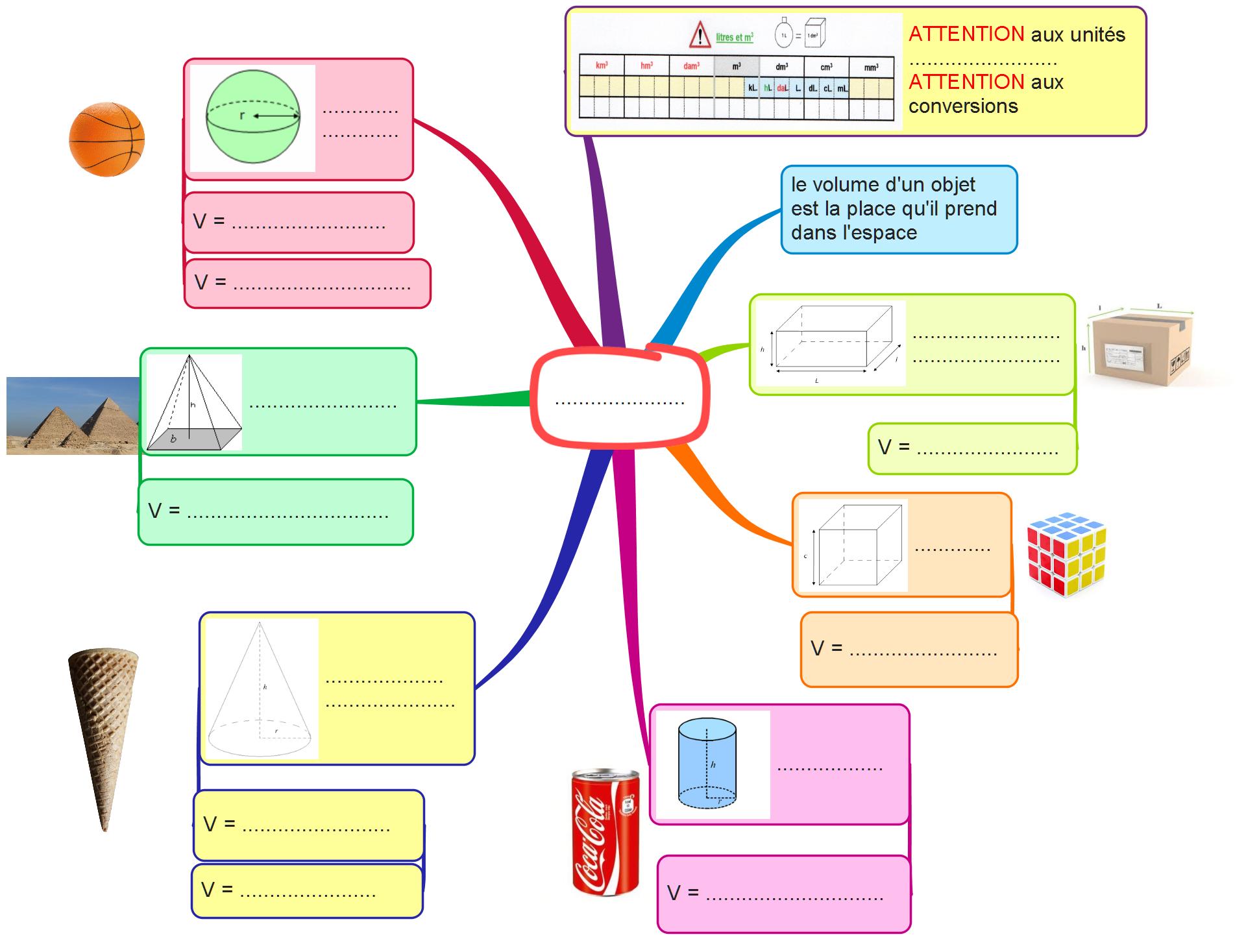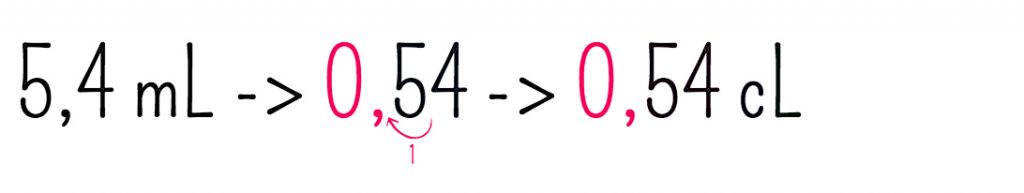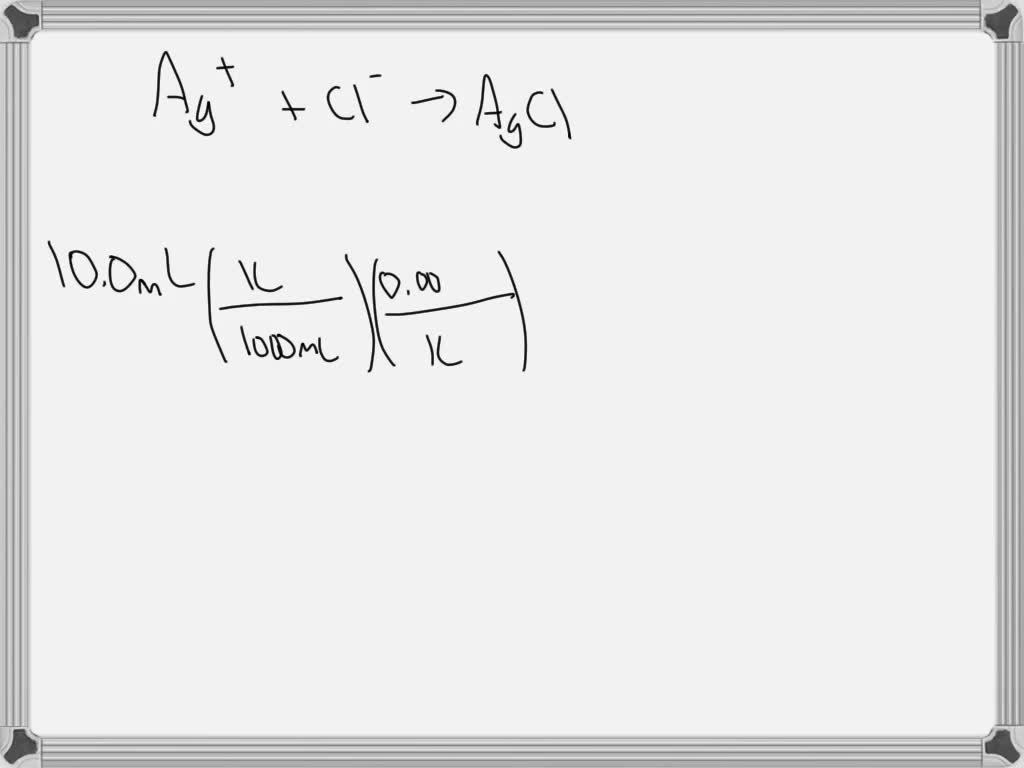
SOLVED: A volume of 10.0 mL of a 0.00350 M solution of Cl– ions are reacted with a 0.500 M solution of AgNO3. What is the maximum mass of AgCl that precipitates?
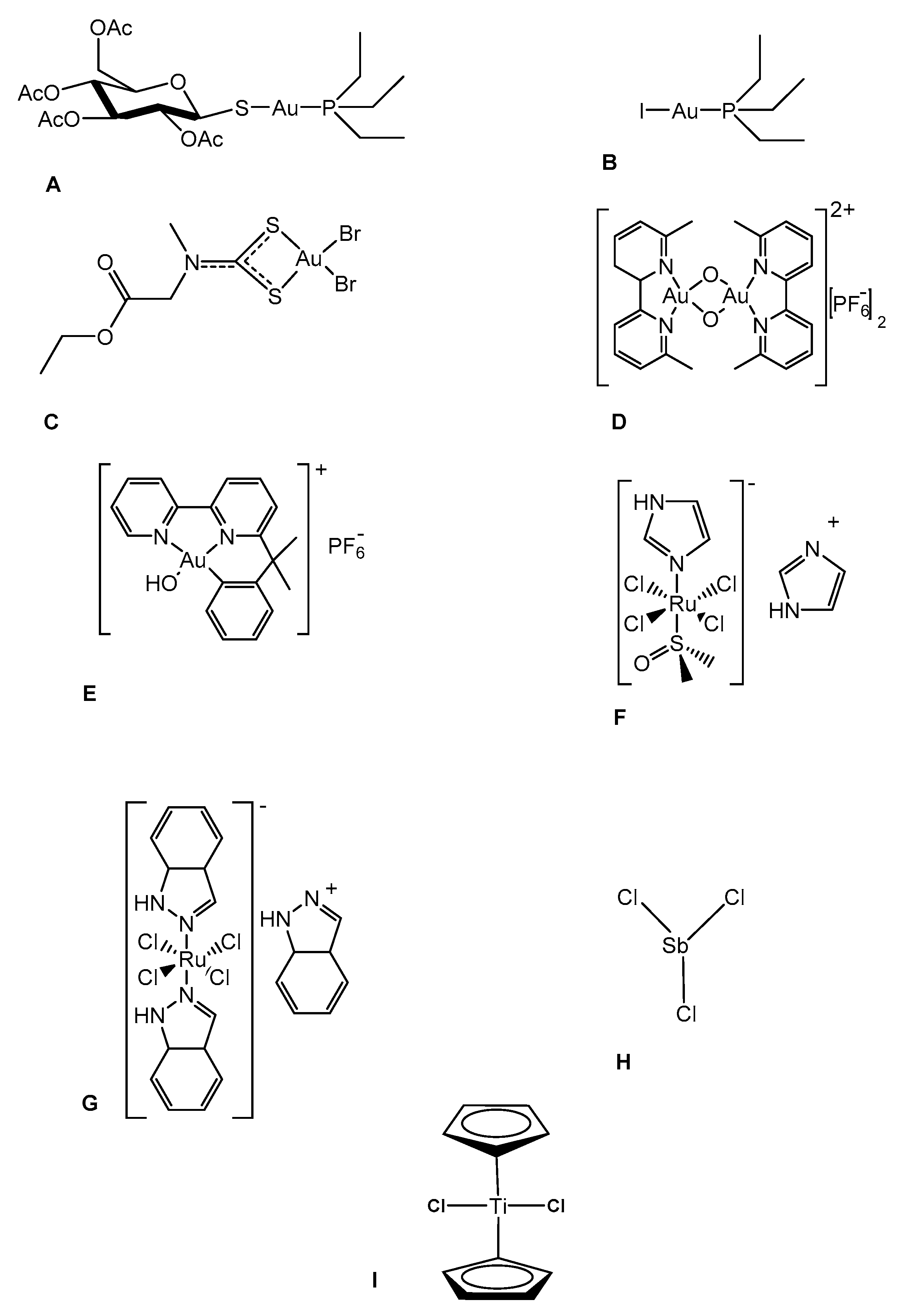
Biomolecules | Free Full-Text | In Vitro Anti-SARS-CoV-2 Activity of Selected Metal Compounds and Potential Molecular Basis for Their Actions Based on Computational Study

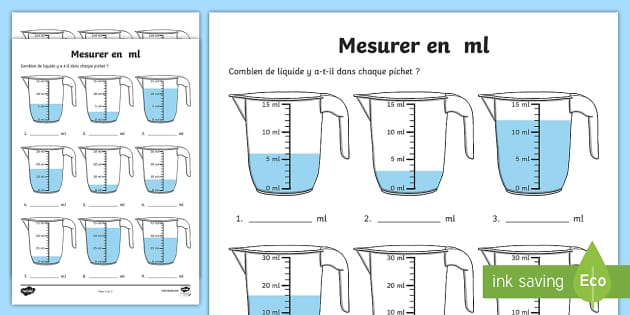
9782803448395: exercices de calcul 23. poids et mesures (9-10 a.) - De Smedt/Jansen: 2803448394 - AbeBooks

SOLVED: A 450.0 -mL sample of a 0.257-M solution of silver nitrate is mixed with 400.0 mL of 0.200M calcium chloride. What is the concentration of Cl^- in solution after the reaction

Expanding the Polymethine Paradigm: Evidence for the Contribution of a Bis-Dipolar Electronic Structure | The Journal of Physical Chemistry A

SOLVED: Calculate the approximate volume (in mL) of 0.1 M silver nitrate (AgNO3) that will be needed to precipitate the chloride from a 0.1156g chloride salt sample that is 55% weight chloride