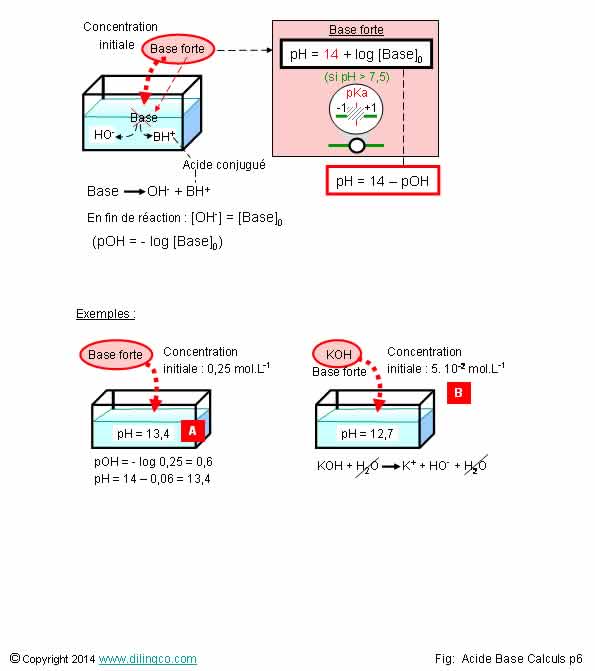
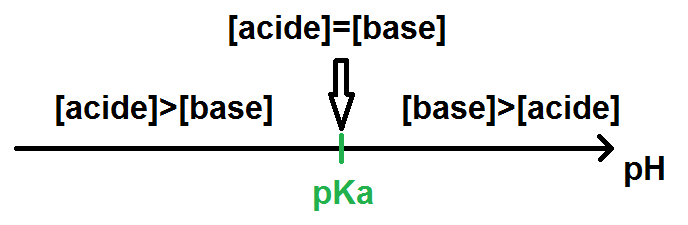
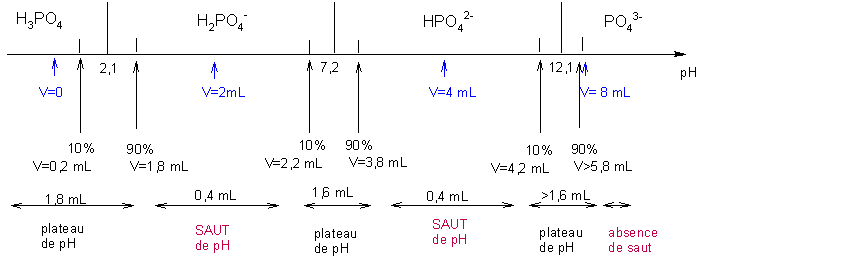
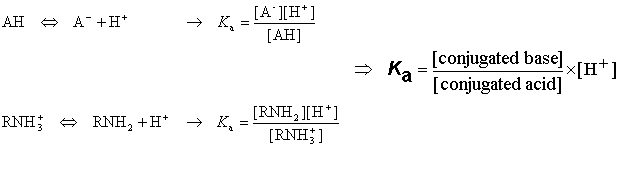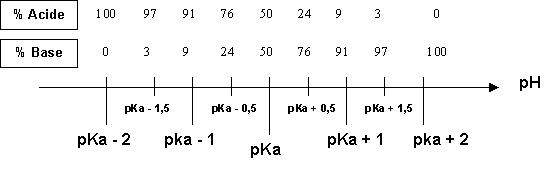![PH = pKa + log [CH3COO-] = = [CH3COOH] = = = × = = 2 Donc pKa = pH – log 2 = 4,7. - ppt video online télécharger PH = pKa + log [CH3COO-] = = [CH3COOH] = = = × = = 2 Donc pKa = pH – log 2 = 4,7. - ppt video online télécharger](https://slideplayer.fr/slide/1711024/7/images/2/pH+%3D+pKa+%2B+log+%5BCH3COO-%5D+%3D+%3D+%5BCH3COOH%5D+%3D+%3D+%3D+%C3%97+%3D+%3D+2..jpg)
PH = pKa + log [CH3COO-] = = [CH3COOH] = = = × = = 2 Donc pKa = pH – log 2 = 4,7. - ppt video online télécharger

Using Atomic Charges to Describe the pKa of Carboxylic Acids | Journal of Chemical Information and Modeling

Predicting pKa Values of Quinols and Related Aromatic Compounds with Multiple OH Groups | The Journal of Organic Chemistry
![SOLVED:Phenolphthalein has a pKa of 9.7. It is colorless in its acid form and pink in its basic form. For each of the pH values, calculate [In-]/[HIn] and predict the color of SOLVED:Phenolphthalein has a pKa of 9.7. It is colorless in its acid form and pink in its basic form. For each of the pH values, calculate [In-]/[HIn] and predict the color of](https://cdn.numerade.com/previews/6eaf3b36-e3f4-4e6b-941d-93649ba4240b.gif)