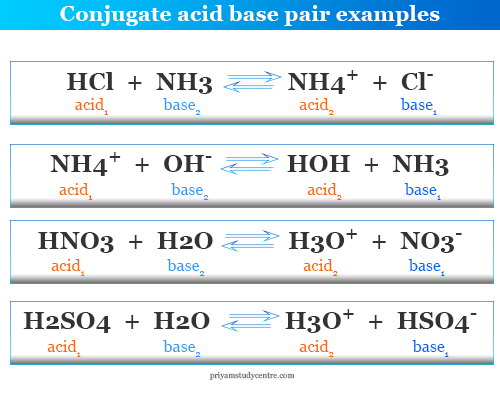
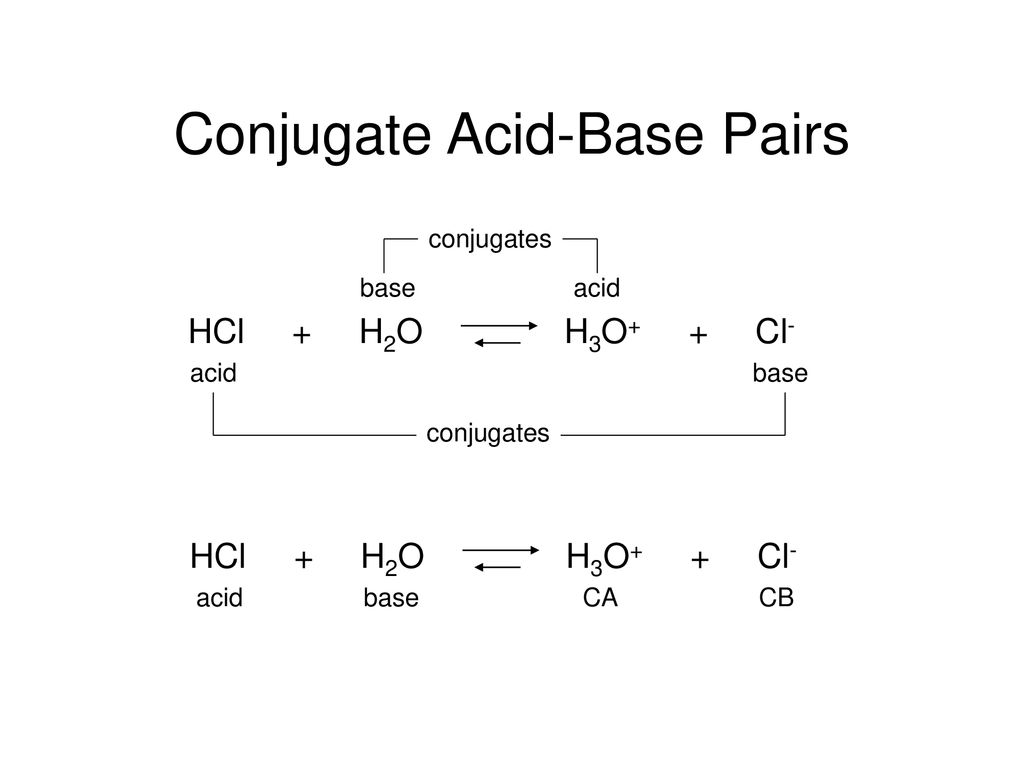
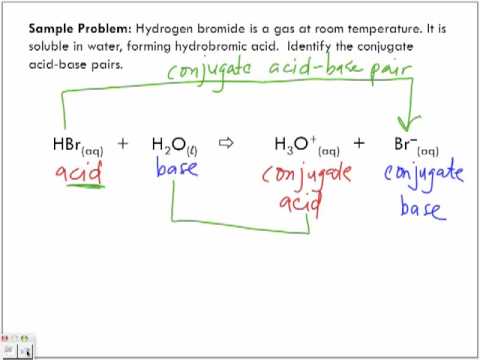
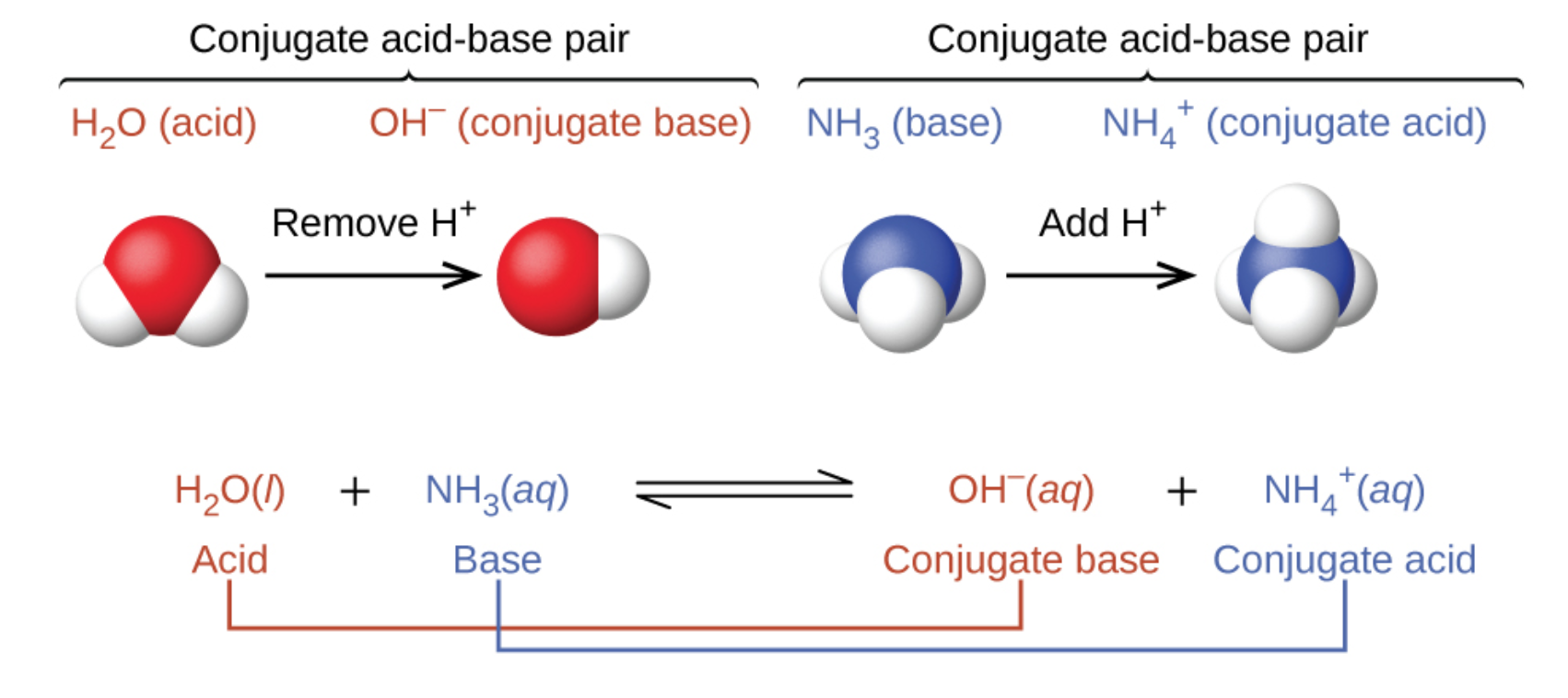
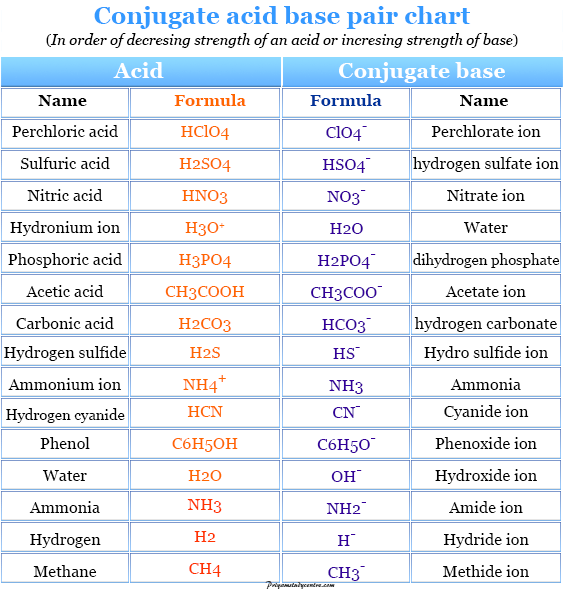
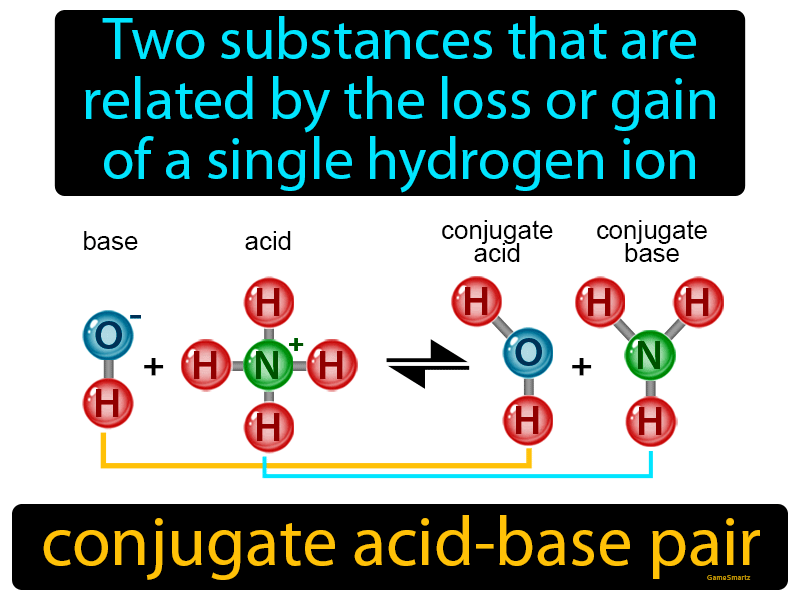
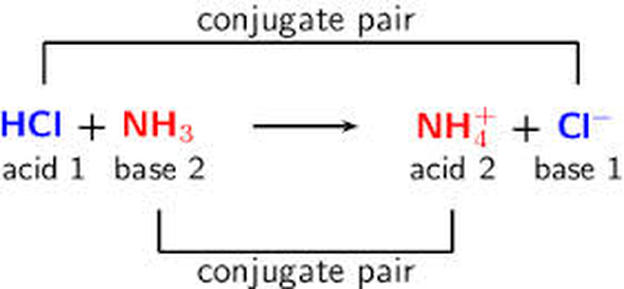
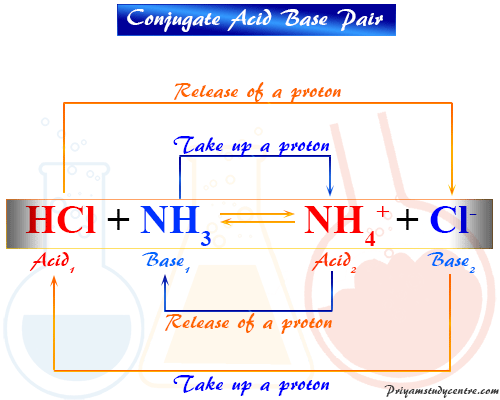
Relative strengths of some common conjugate acid- base pairs, listed... | Download Scientific Diagram

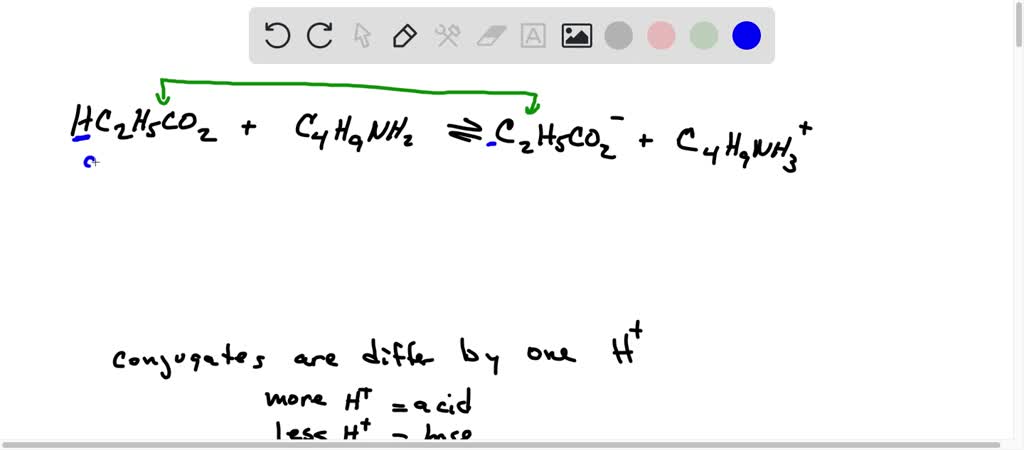
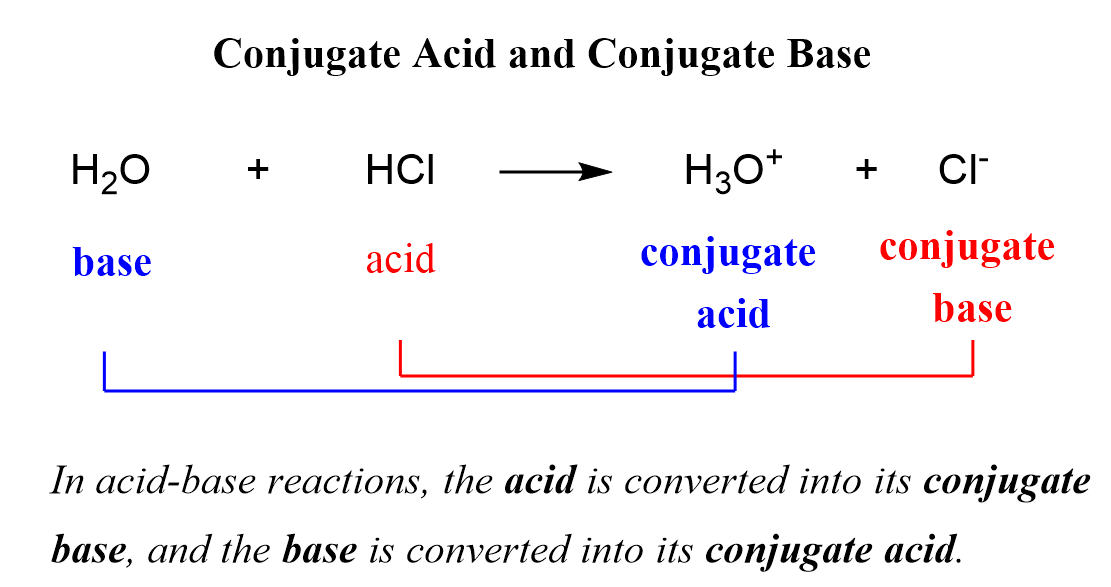
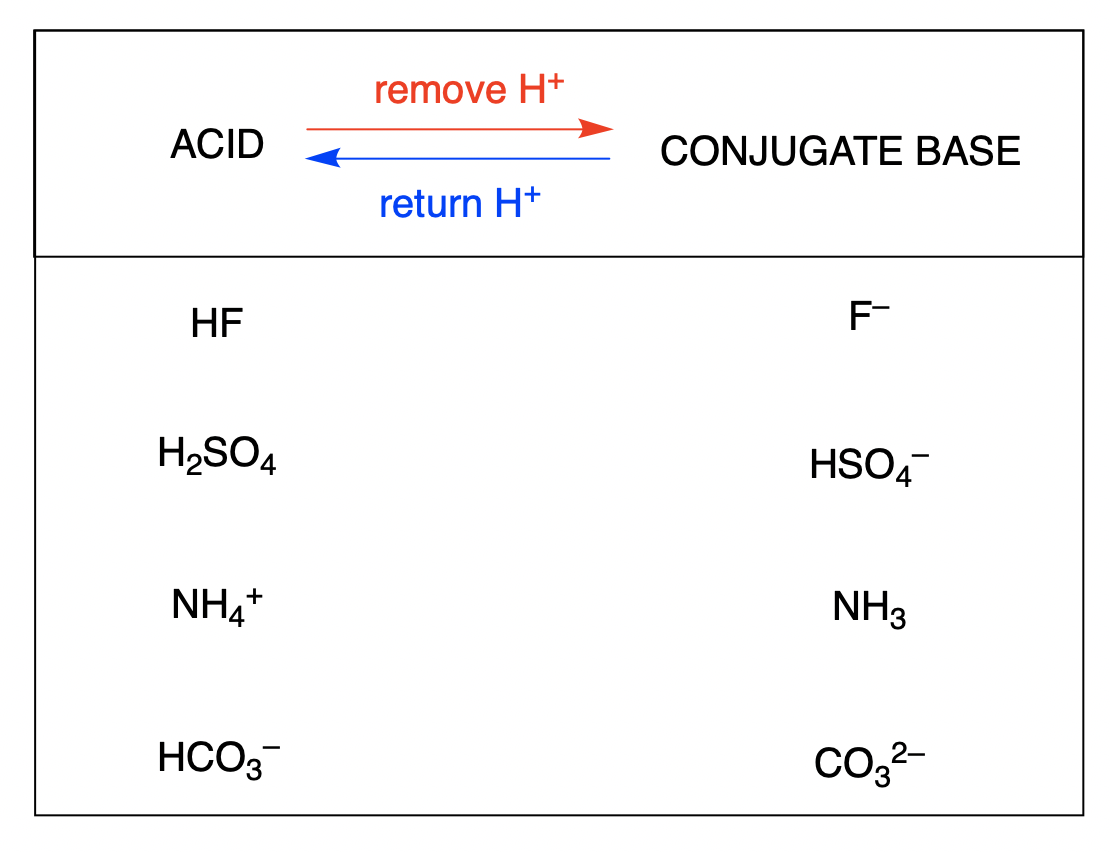
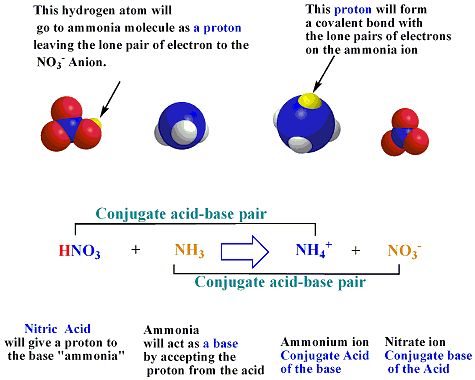
Identify the conjugate acid-base pairs for the reaction (with the acid written first). CN- + H2O = HCN + OH- |CN- / HCN |HCN / CN- |OH- / H2O |H2O / OH- | Homework.Study.com