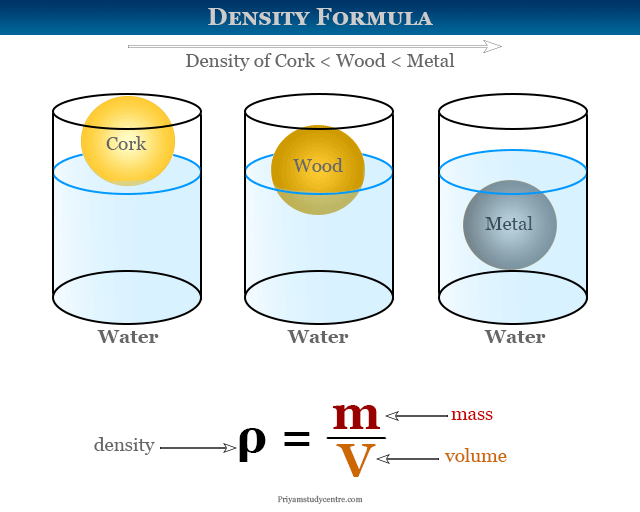
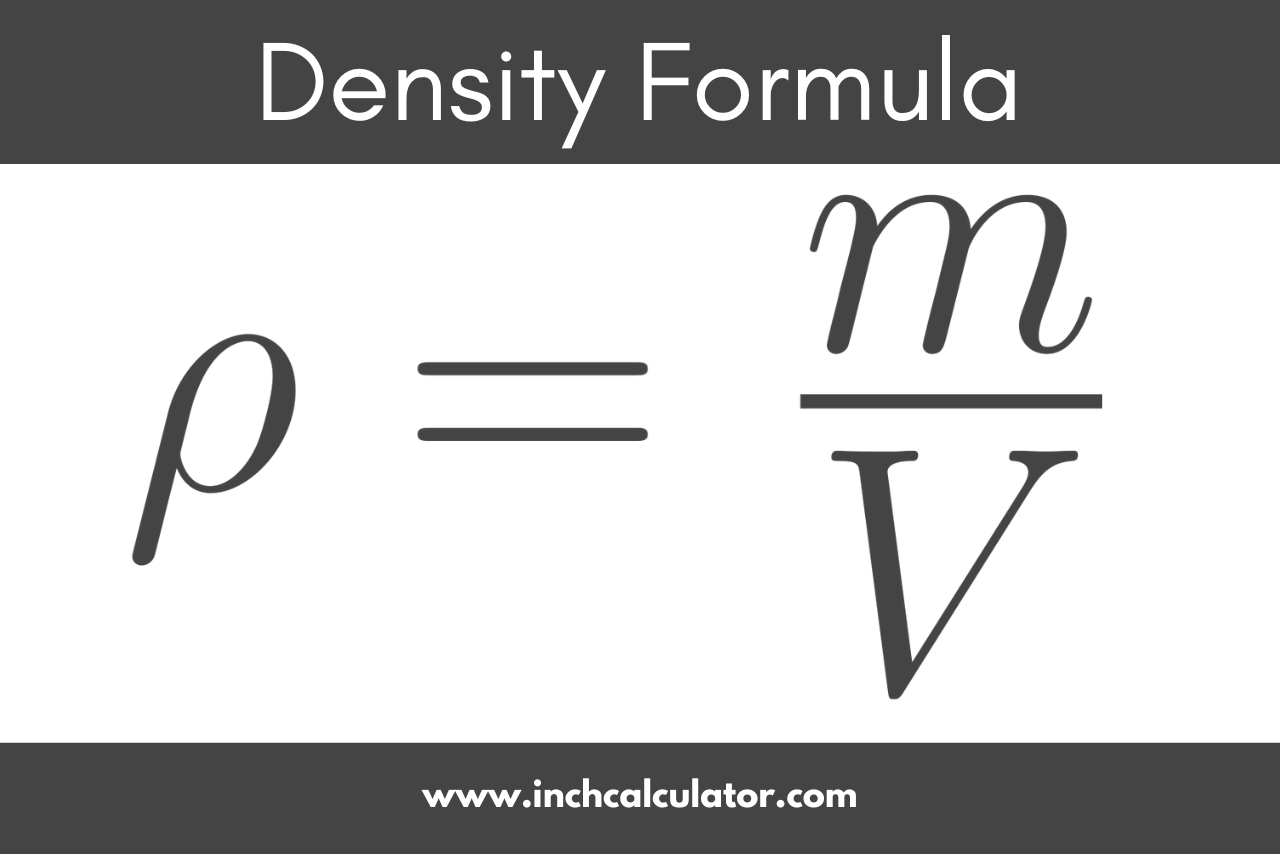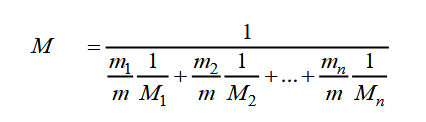An element with density 2.8 g cm^-3 forms a fcc unit cell with edge length 4 × 10^-8 cm . Calculate the molar mass of the element? (Given NA = 6.022 × 10^23mol^-1 )

SOLVED: For this assignment, I have to calculate Molar Mass by Vapor Density. I was given the measurements and have to find the molar mass by vapor density using that. I tried

Molarity, Molality, Volume & Mass Percent, Mole Fraction & Density - Solution Concentration Problems - YouTube