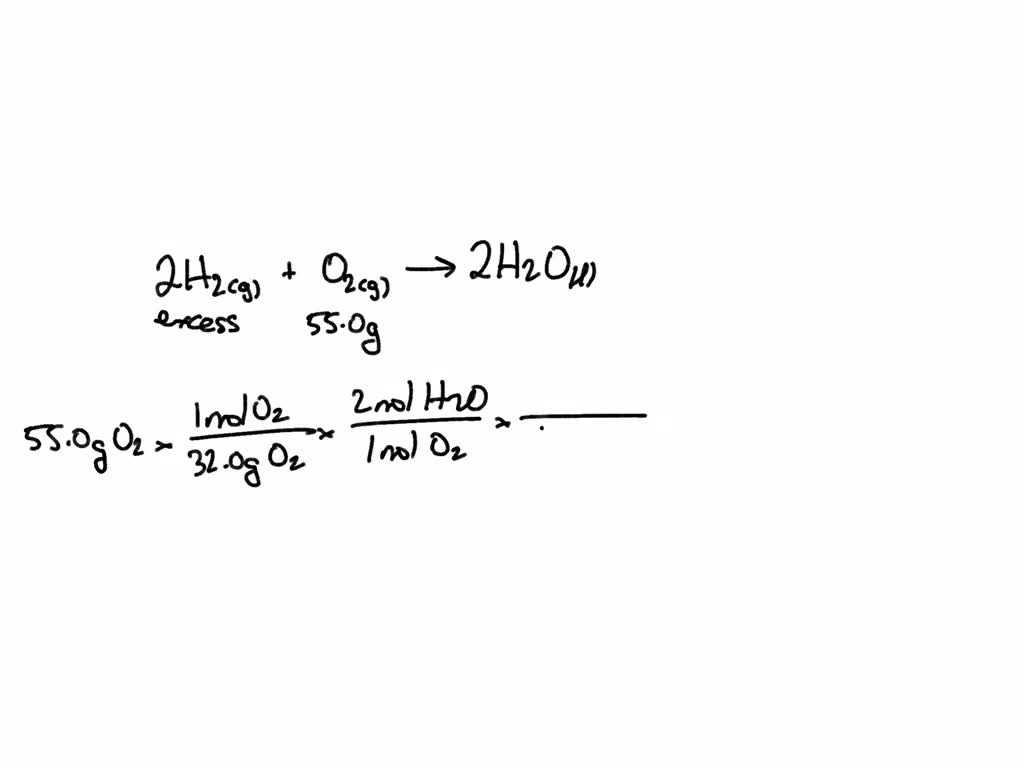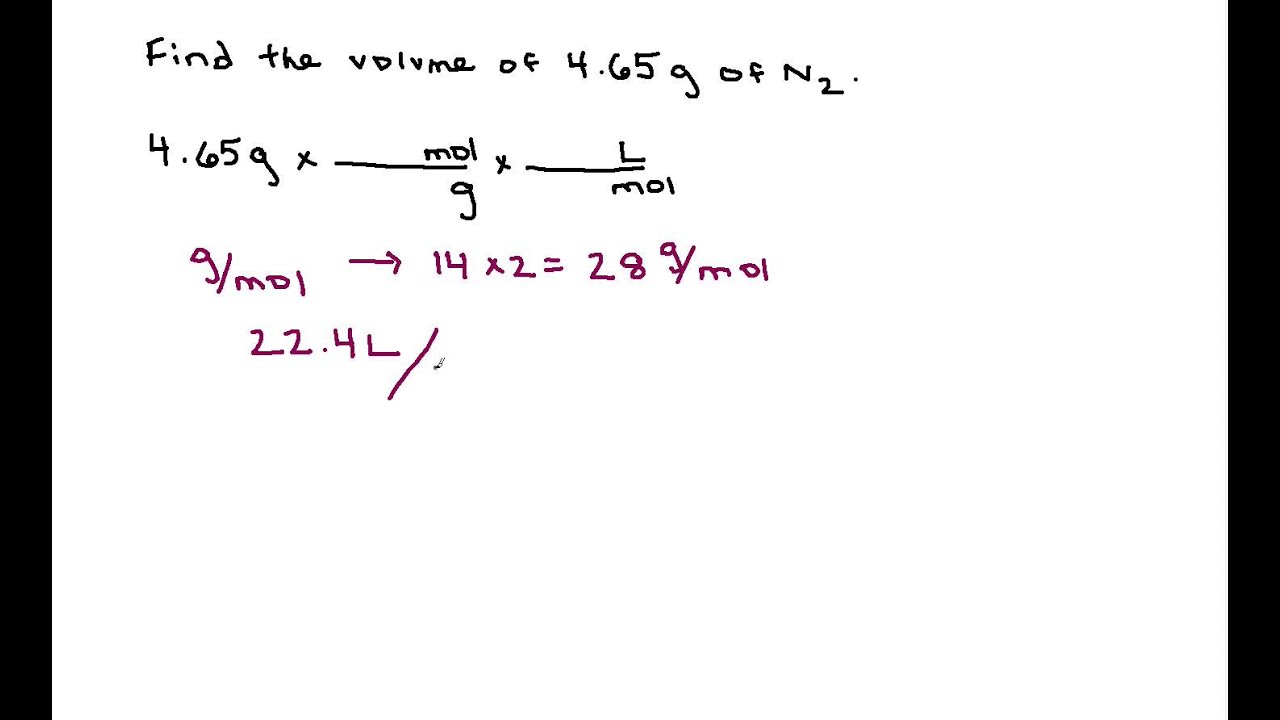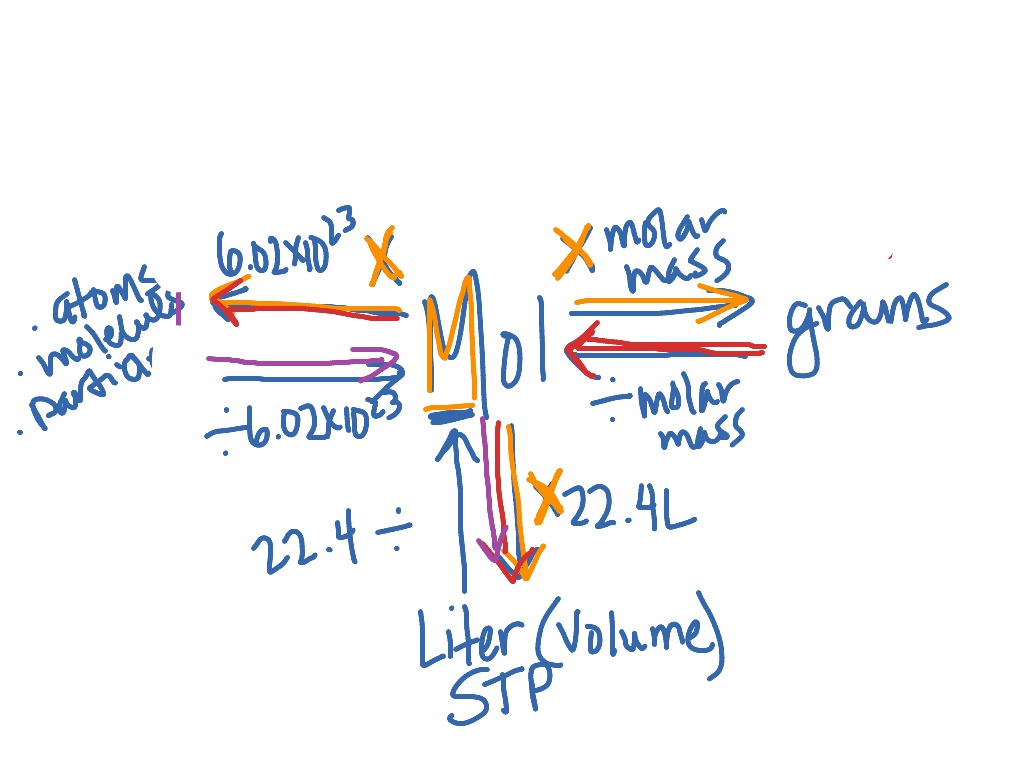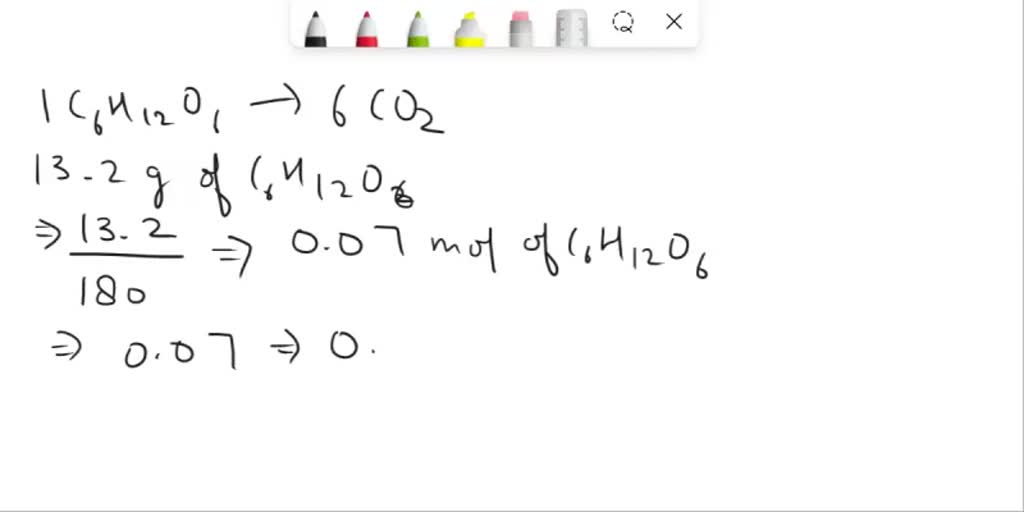
SOLVED: C6H12O6(s) + 6O2(g) –> 6H2O(g) + 6CO2(g) How many liters of CO2 are produced when you start with 13.2 grams of glucose (C6H12O6) in excess oxygen at STP? 0.439 liters CO2
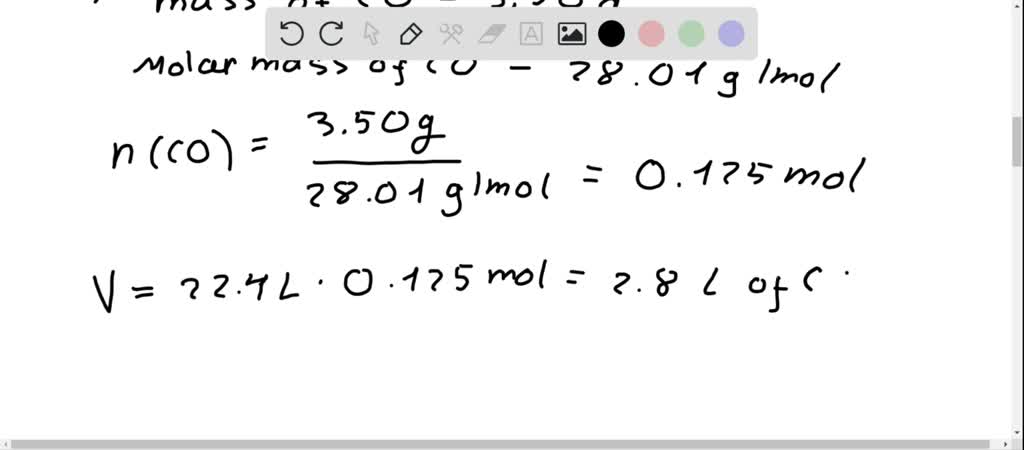
SOLVED:Find the volume, in liters, of each of the following at STP. a. 8.00 g O2 b. 3.50 g CO c. 0.0170 g H2 S d. 2.25 ×10^5 kg NH3

What volume, in liters, of 0.023 M NH4NO3 solution can be made using 25 g NH4NO3 The molar mass of NH4NO3 - Brainly.com