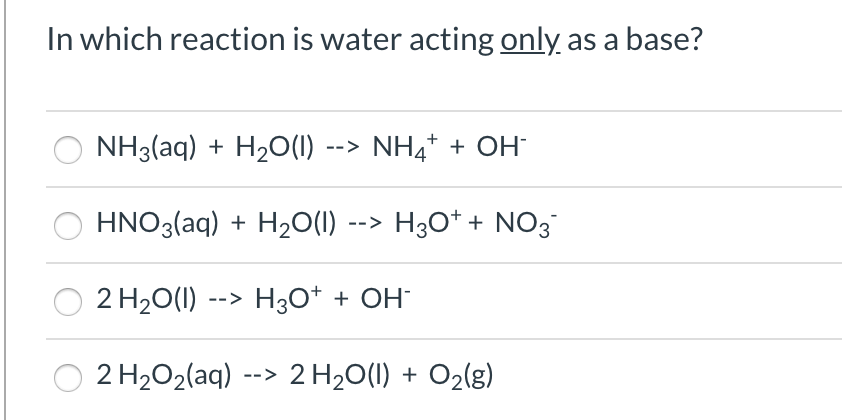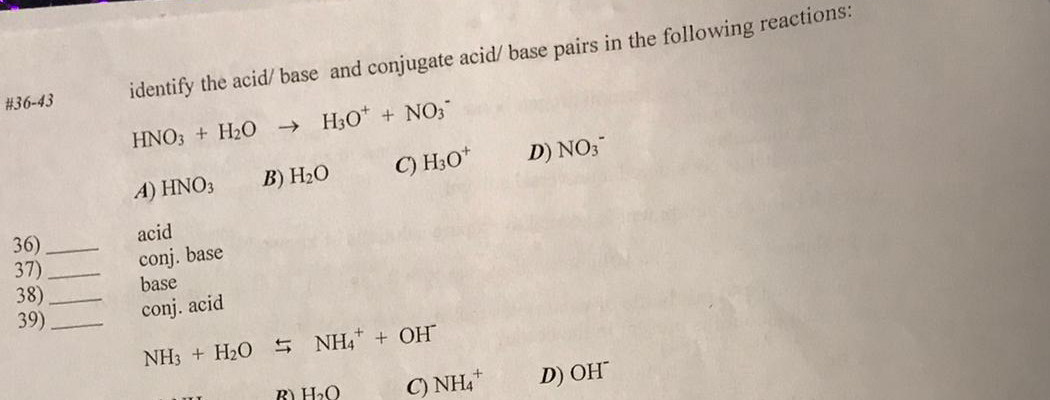Question Video: Calculating the Concentration of Nitric Acid via Titrating against a Known Volume of Potassium Hydroxide | Nagwa

Nucleation of Mixed Nitric Acid–Water Ice Nanoparticles in Molecular Beams that Starts with a HNO3 Molecule | The Journal of Physical Chemistry Letters

Vibrational Signatures of HNO3 Acidity When Complexed with Microhydrated Alkali Metal Ions, M+·(HNO3)(H2O)n=5 (M = Li, K, Na, Rb, Cs), at 20 K | The Journal of Physical Chemistry A

In which of the following pair of reactions first reaction is spontaneous while second reaction is non spontaneous?

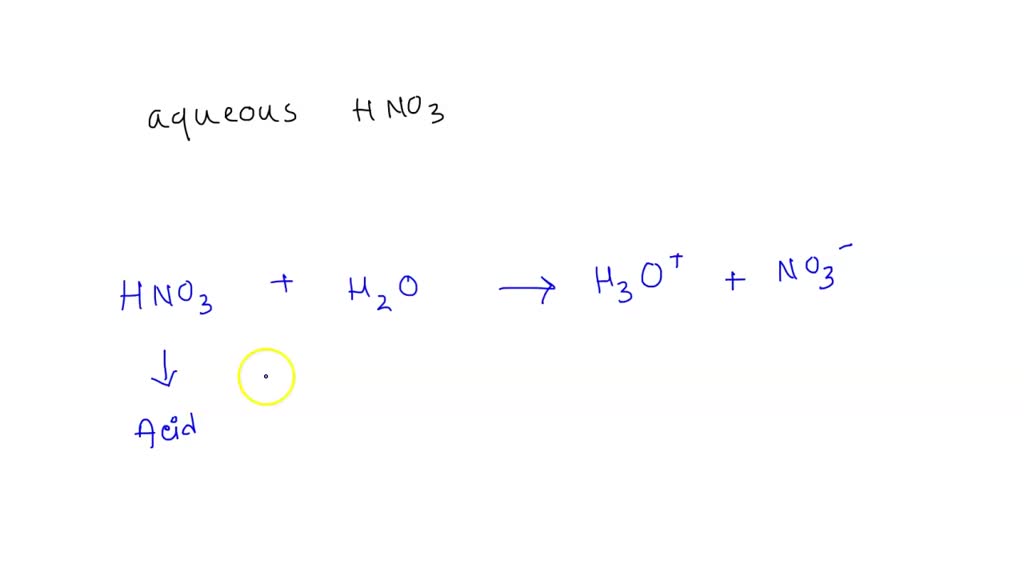
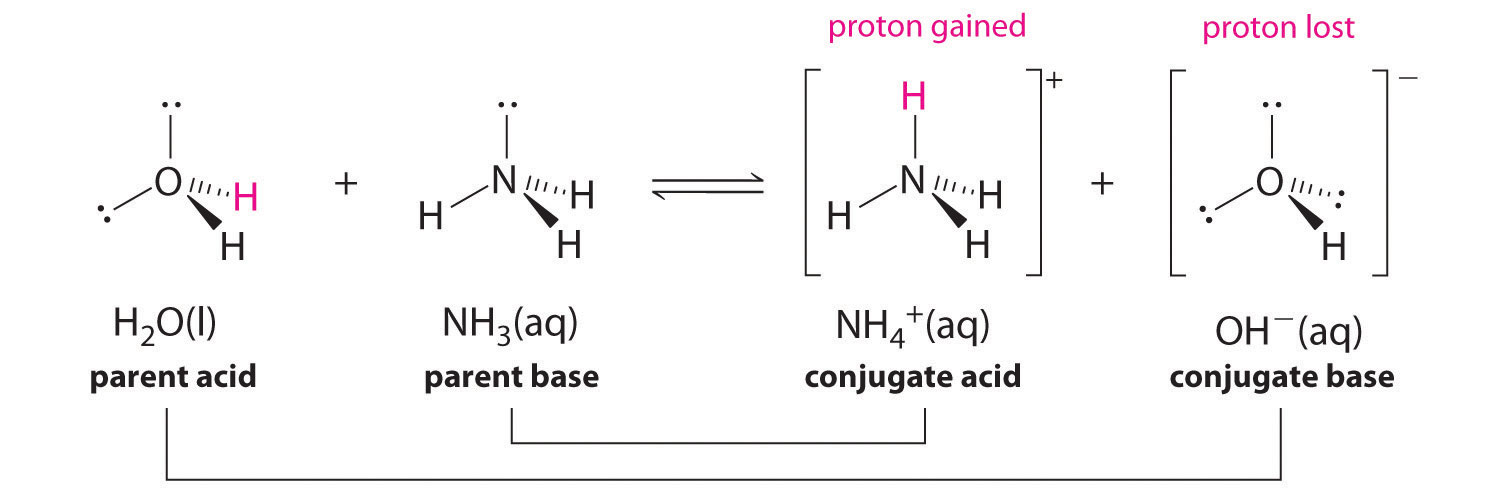
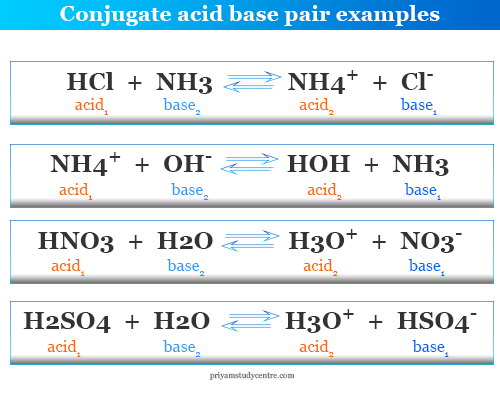
SOLVED: Copy the following equation, and label the Bronsted-Lowry acid, its conjugate base, the Bronsted-Lowry base, and its conjugate acid. HNO3 + H2O → H30* + NO3

Identify the acid, base, conjugate acid, and conjugate base in the following reaction. HNO3 + NH3 + NO3 + - Brainly.com

Sort out the conjugate pairs of acid and bases in the following reactions : HNO3(aq) +H2O (l)hArr H3O^(+) (aq) +NO3^(-) (aq)