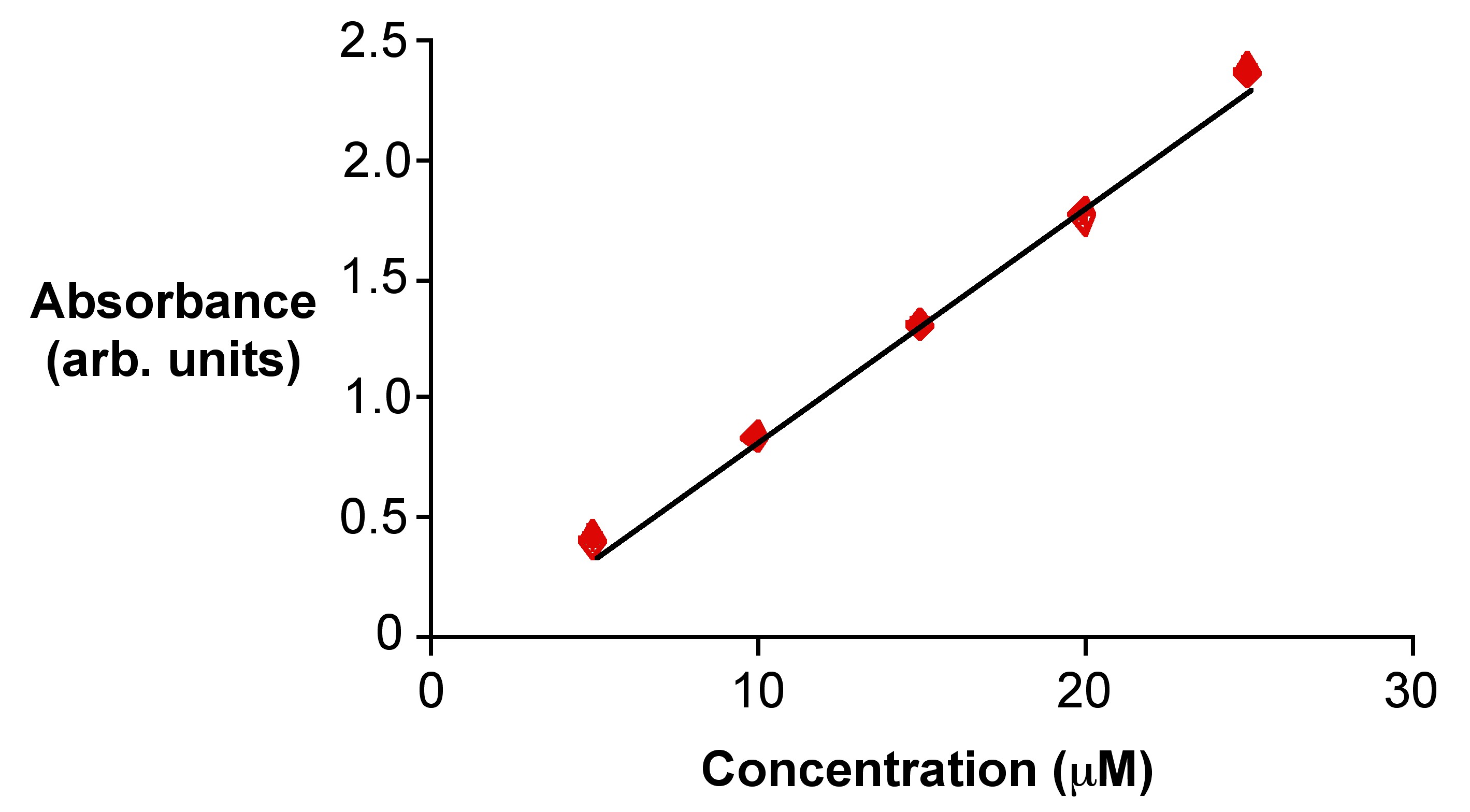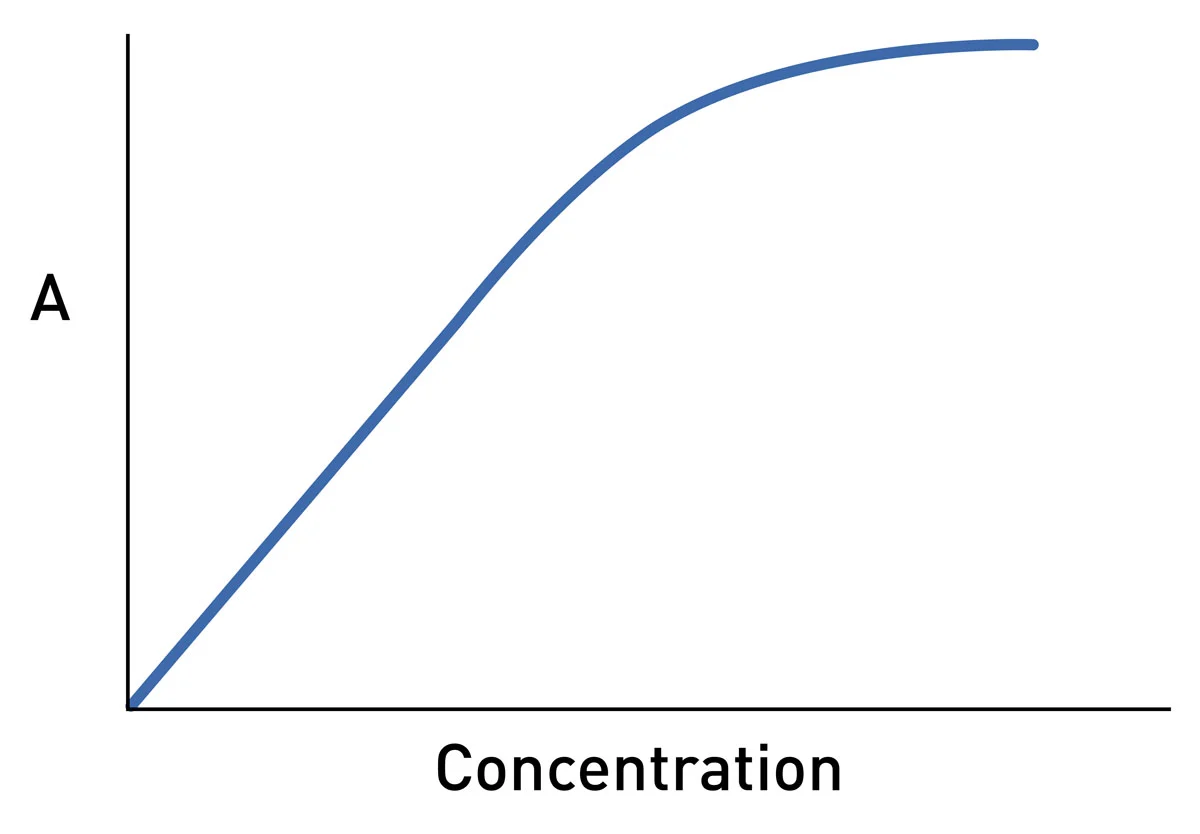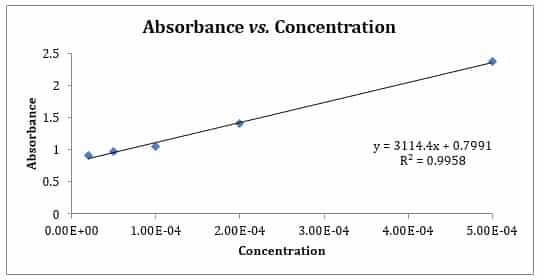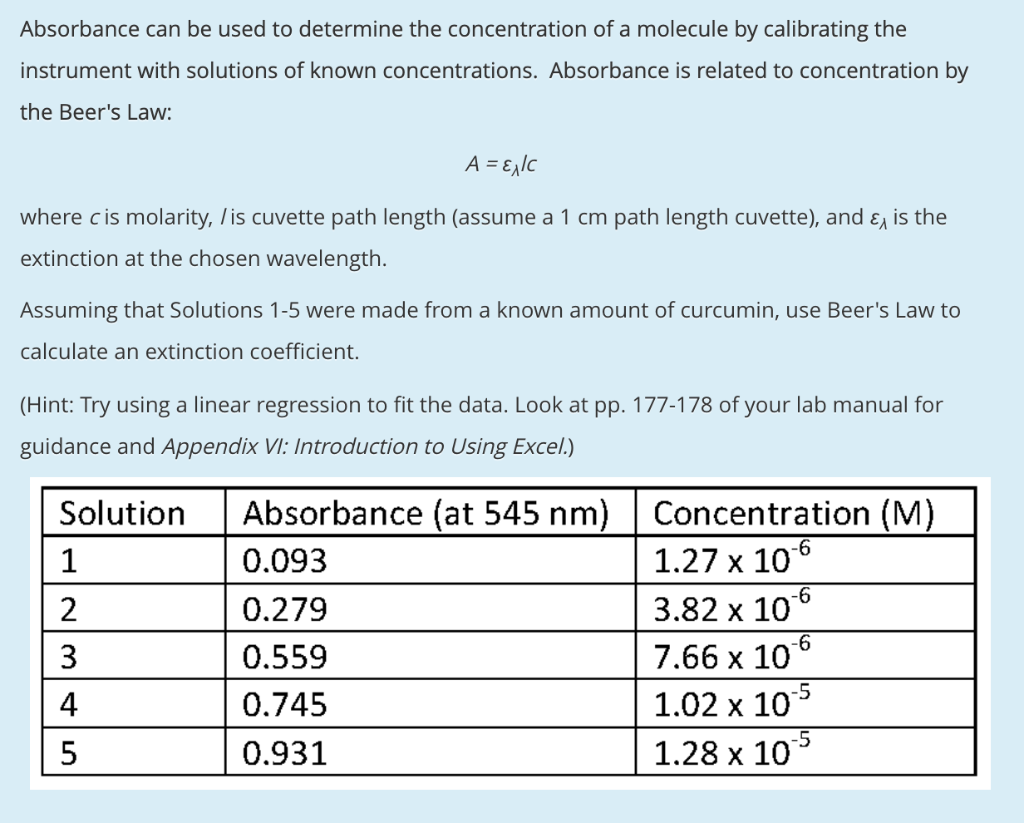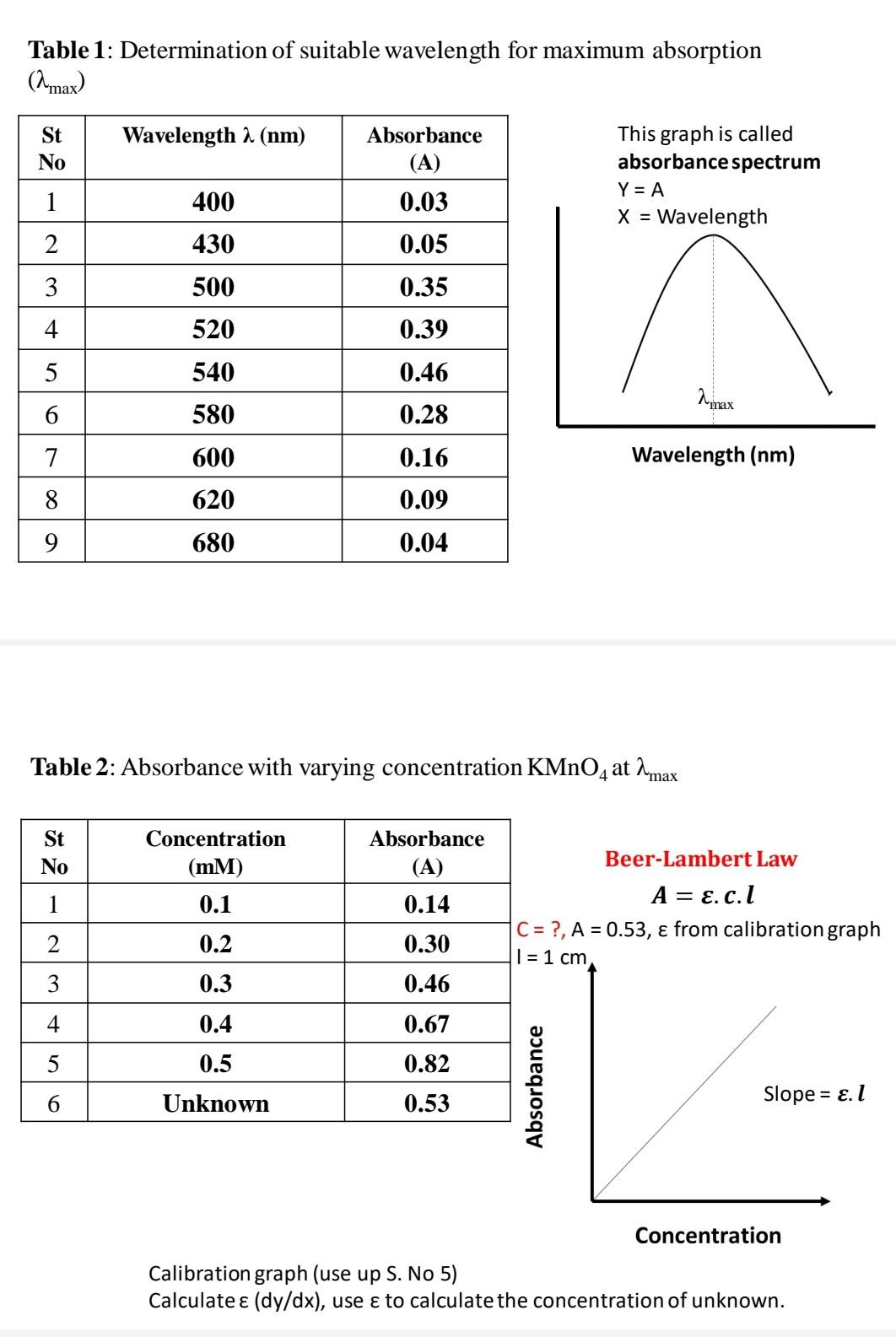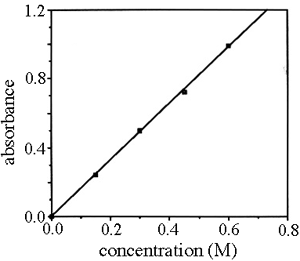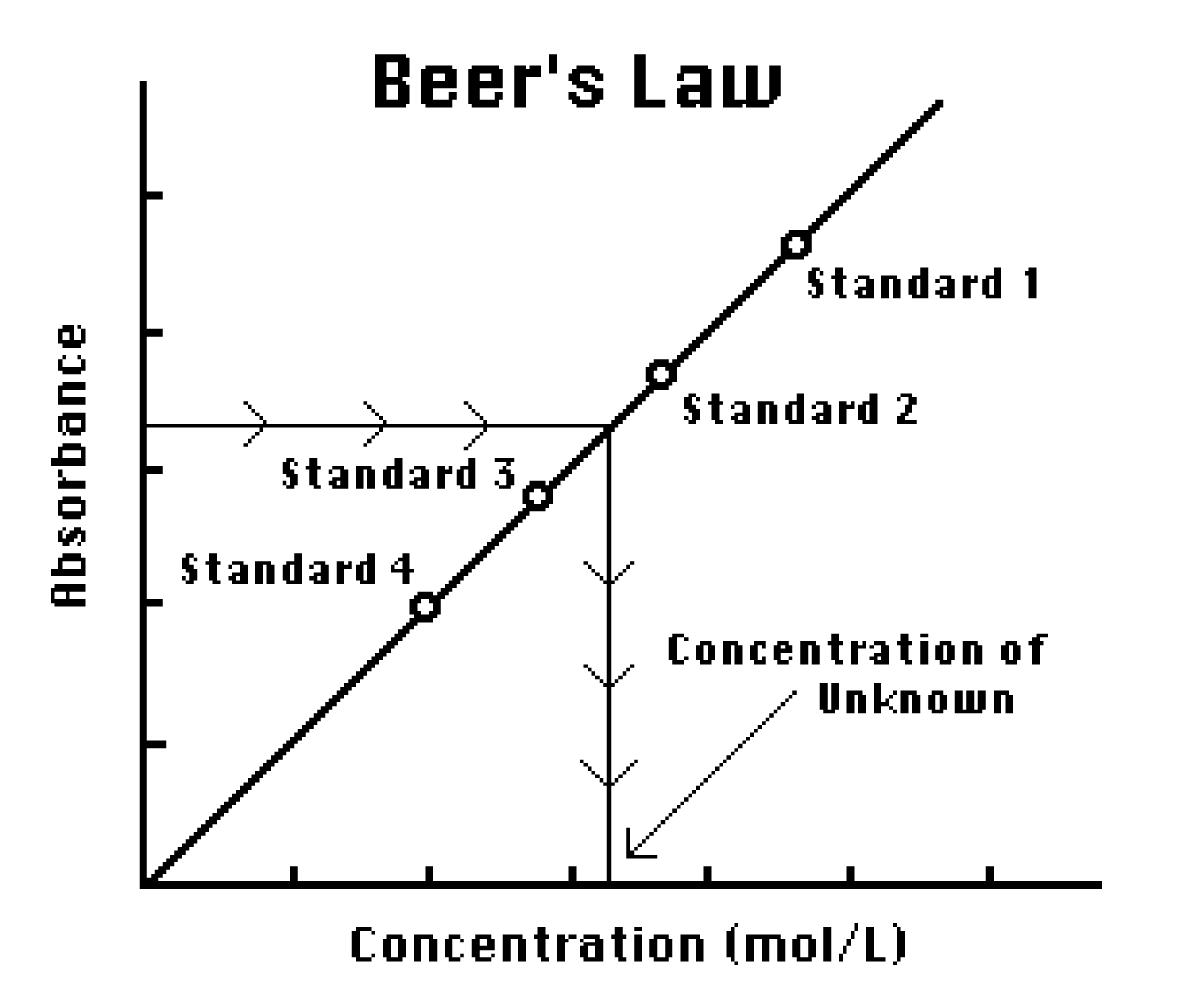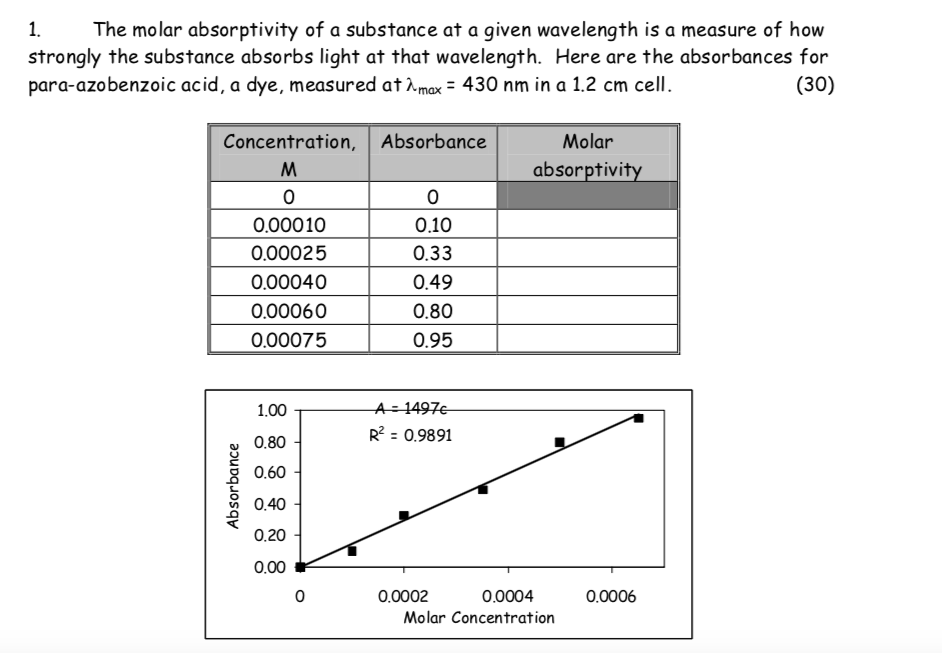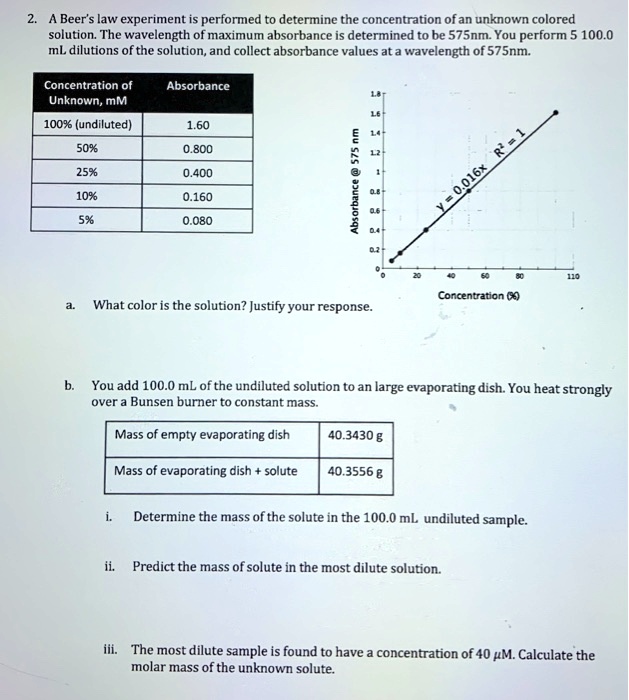
SOLVED: Beer'law experiment is performed to determine the concentration ofan unknown colored solution. The wavelength of maximum absorbance is determined to be 575nm. You perform 5 100.0 mL dilutions of the solution;

UV-Vis Spectrophotometry – Easy and Quick Quantification of Nucleic Acids - Eppendorf Handling Solutions

SOLVED: Problems Explain the difference between transmittance absorbance; and molar absorptivity. Which one is proportional to concentration? The absorbance of a 2.3x 10 ' M solution of a compound is 0.822 at

Worked example: Calculating concentration using the Beer–Lambert law | AP Chemistry | Khan Academy - YouTube

SOLVED: Using the data below for the serially diluted samples, determine the concentration of your purified calf thymus DNA Concentration SOnglul 2Sng/ul 12.Sng/ul 6.25ng/ul Absorbance 0.978 0.521 0.253 0.125 Construct a standard

Absorbance Measurements – the Quick Way to Determine Sample Concentration - Eppendorf Handling Solutions