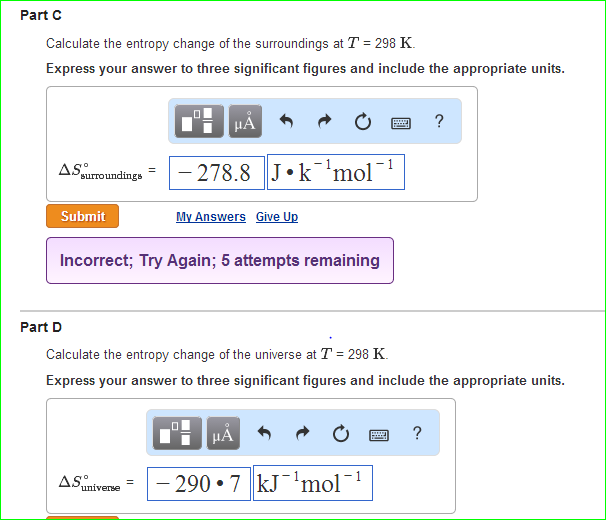
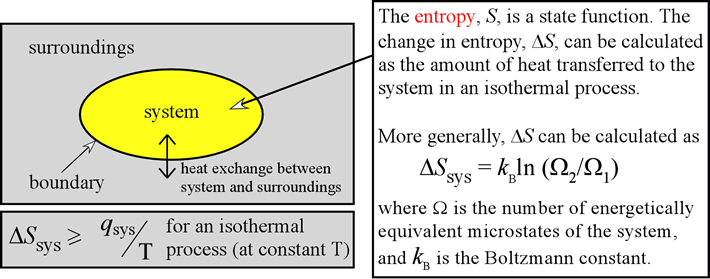
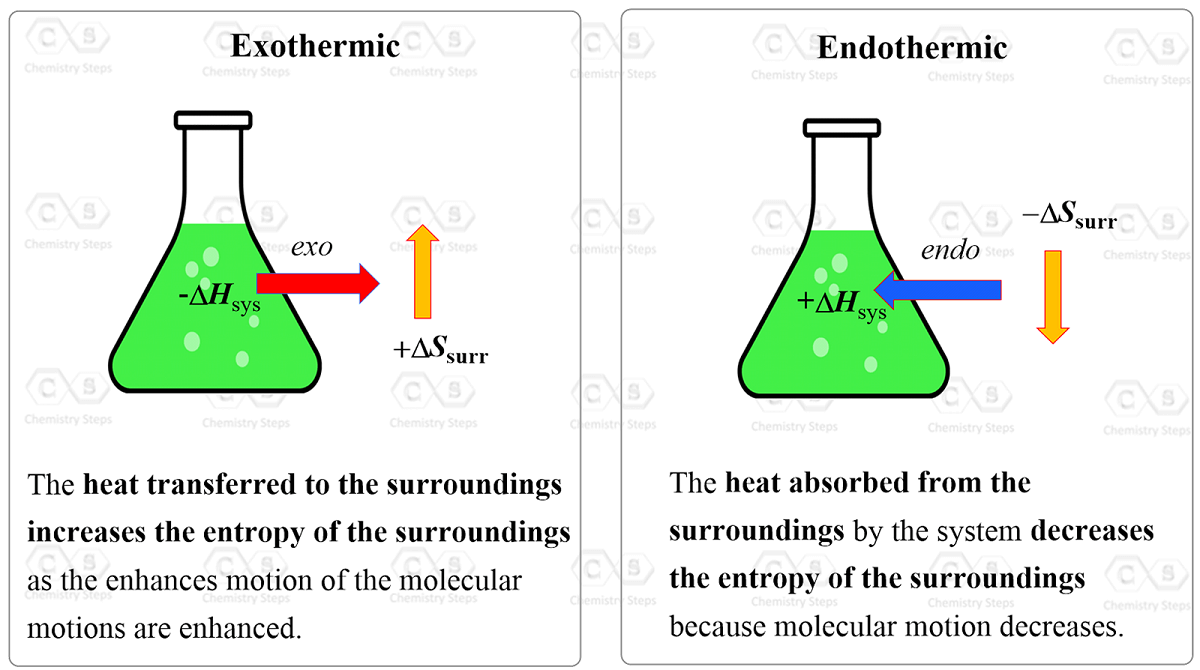
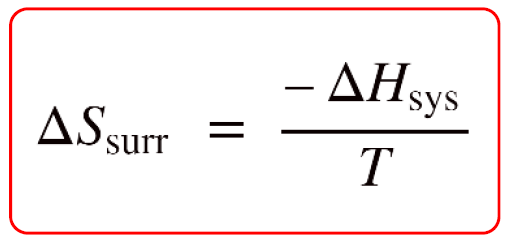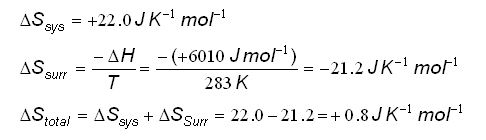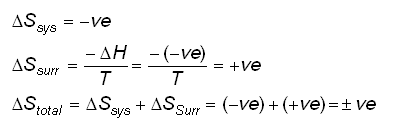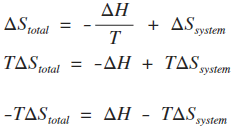Calculate ΔS univ (in J/K) for the chemical reaction: C(graphite) + 2H2(g)→CH4(g);ΔH300^o = - 75.0kJ . The standard entropies of C(graphite),H2(g),CH4(g) are 6.0,130.6 and 186.2J/K - mol , respectively.
Calculate the entropy change in the system. and in the surroundings and the total entropy change in the universe when during - Sarthaks eConnect | Largest Online Education Community