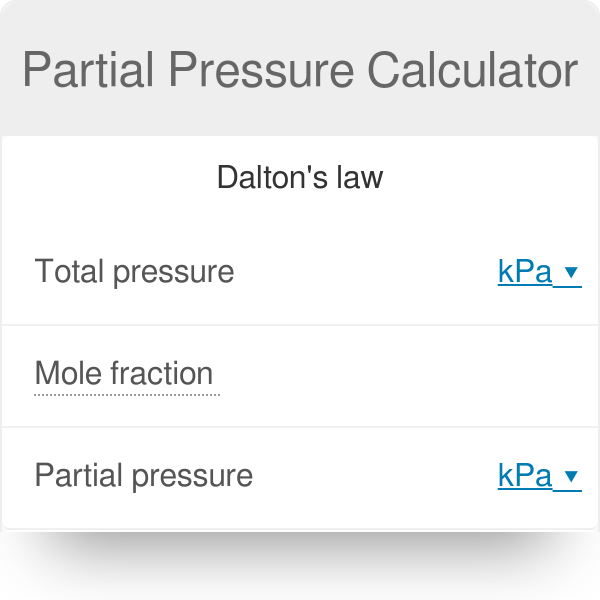
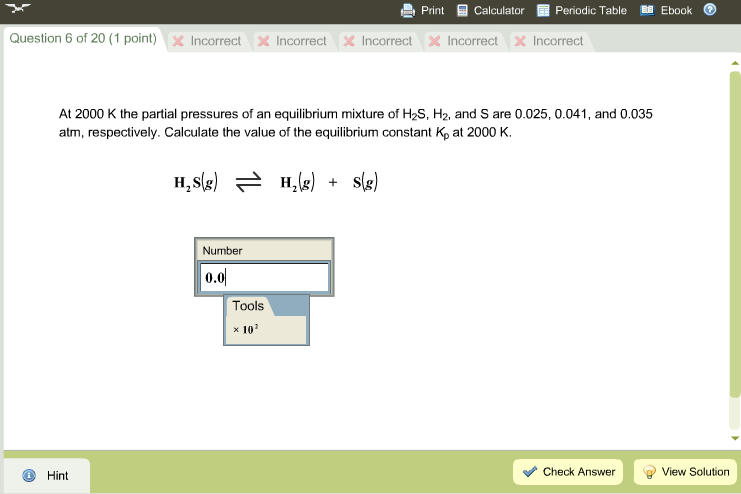
At 87^(@)C , the following equilibrium is established H(2) (g)+S(s)hArrH(2)S(g), K(p)=7xx10^(-2) If 0.50 mole of hydrogen and 1.0 mole of sulphur are heated to 87^(@)C calculate partial pressure of H2S at eq.
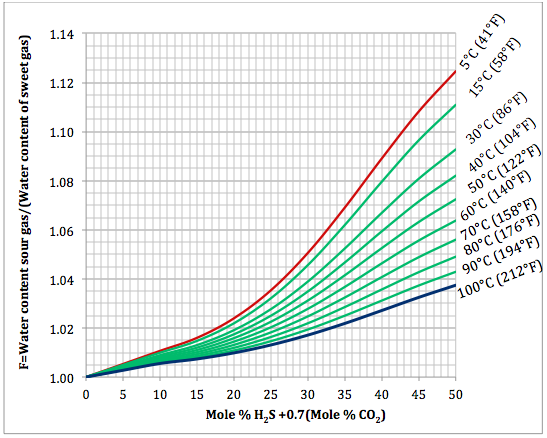
Estimating Sour Gas Water Content by New Correlations and Simplified Charts | Campbell Tip of the Month
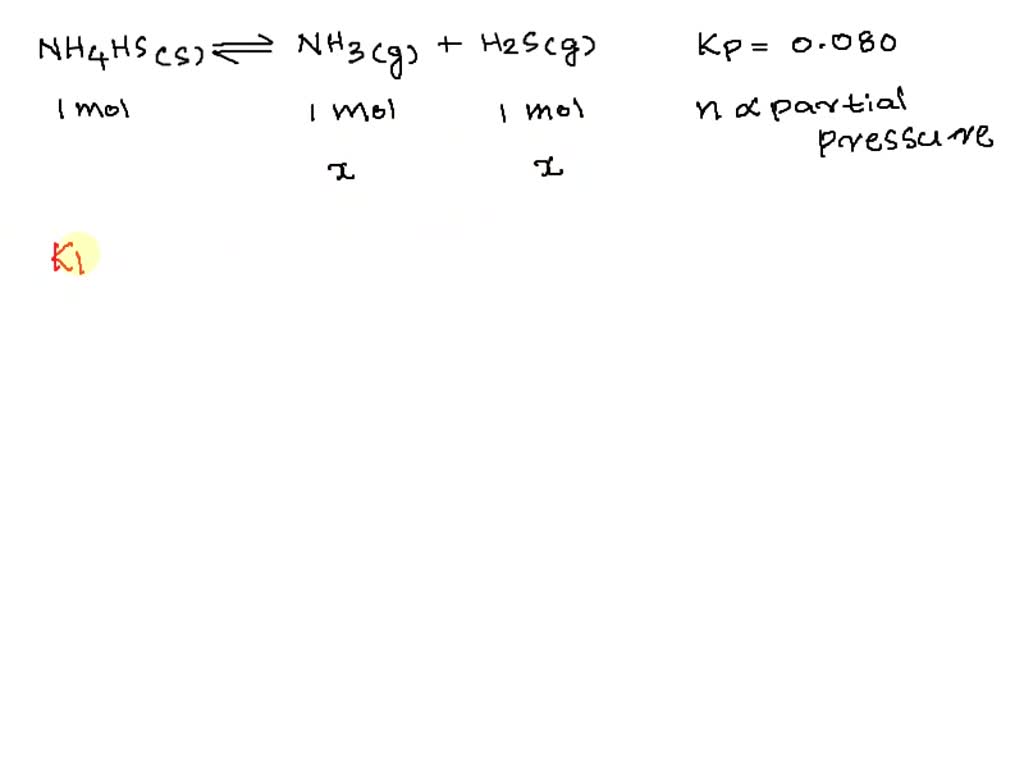
SOLVED: 20) At 24*C, K = 0.080 for the equilibrium: NH4HS (s) NH3 (g) + H2S (g) A sample of solid NH4HS is placed in a closed vessel and allowed to equilibrate.

H2S , a toxic gas with a rotten egg - like smell, is used for the qualitative analysis. If the solubility of H2S in the water at STP is 0.195 m, then
Effect of H 2 S gas concentration in the H 2 S/CO 2 mixture at total... | Download Scientific Diagram
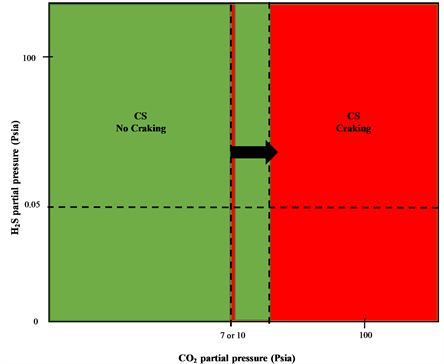
Sulfide Stress Cracking Assessment of Carbon Steel Welding with High Content of H2S and CO2 at High Temperature: A Case Study

Equilibrium H2S partial pressures as a function of liquid loading and... | Download Scientific Diagram

Use the following vapor pressure diagram to estimate the partial pressure of acetone. | Homework.Study.com