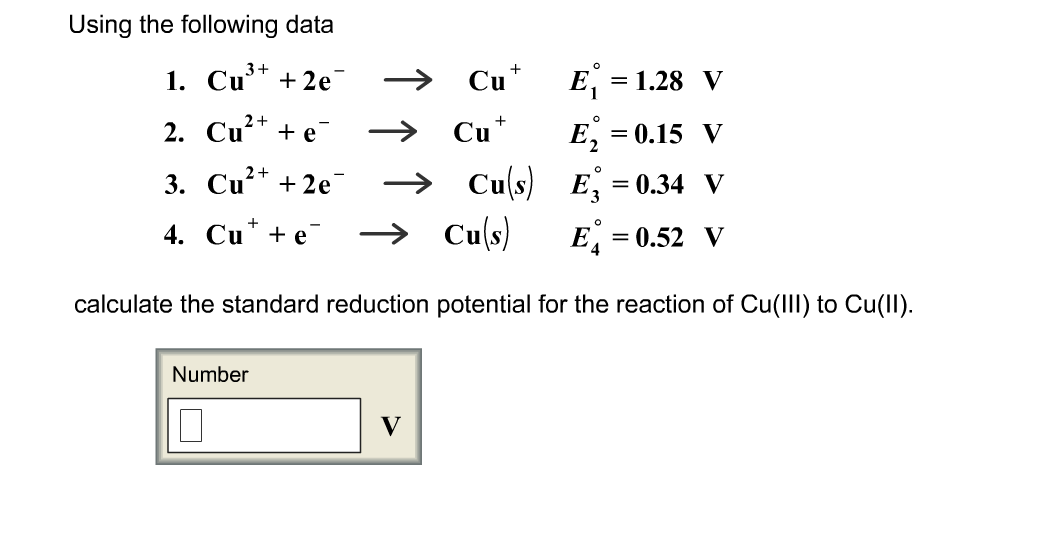
equilibrium - Calculate the cathode electrode potential in this redox reaction - Chemistry Stack Exchange

Standard reduction potentials | Redox reactions and electrochemistry | Chemistry | Khan Academy - YouTube

Calculate the reduction potential of a half cell consisting of a platinum electrode immersed in 2.0 MFe^2 and 0.02 M FE^3 solution. Given E^0Fe^3/Fe^+2 = 0.771 V .
2 The standard half reduction potential of Ag+|Ag is 0.79V is 25^° C. Given the experimental value Ksp=1.5 10* 10 for AgCl, calculate the standard half cell reduction potential for the Ag|AgCl

The standard reduction potential for `Cu^(2+)|Cu` is `+0.34V`. Calculate the reduction potential... - YouTube

OneClass: Given the following half-reactions and their respective standard reduction potentials calcu...

Question Video: Calculating the Standard Cell Potential for a Magnesium/Silver Galvanic Cell | Nagwa

SOLVED: Based on the following standard reduction potentials, Iu2t(aq) + e" Int(aq) =-0.40 V Int(aq) + e- 5 In(s) E =-0.14V calculate the standard reduction potential for the half-reaction Iult(aq) + 2e"