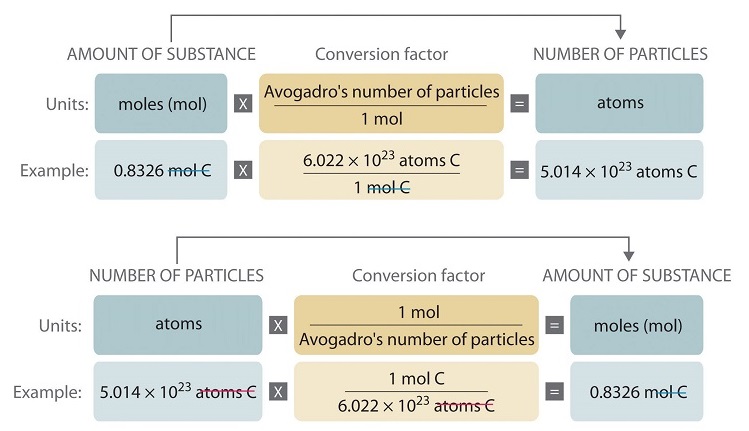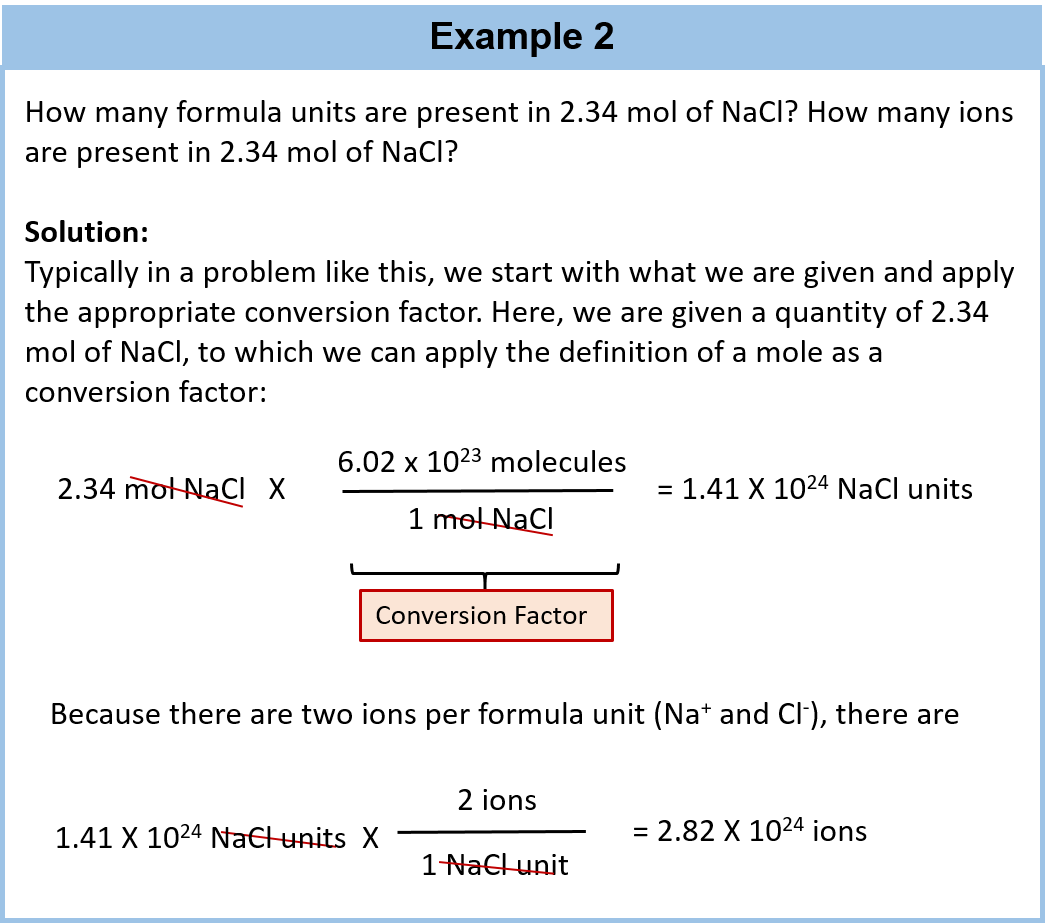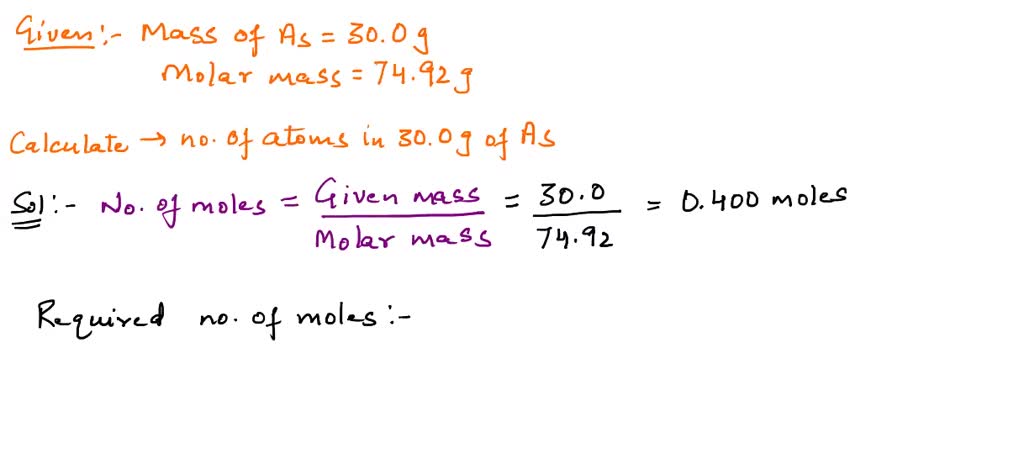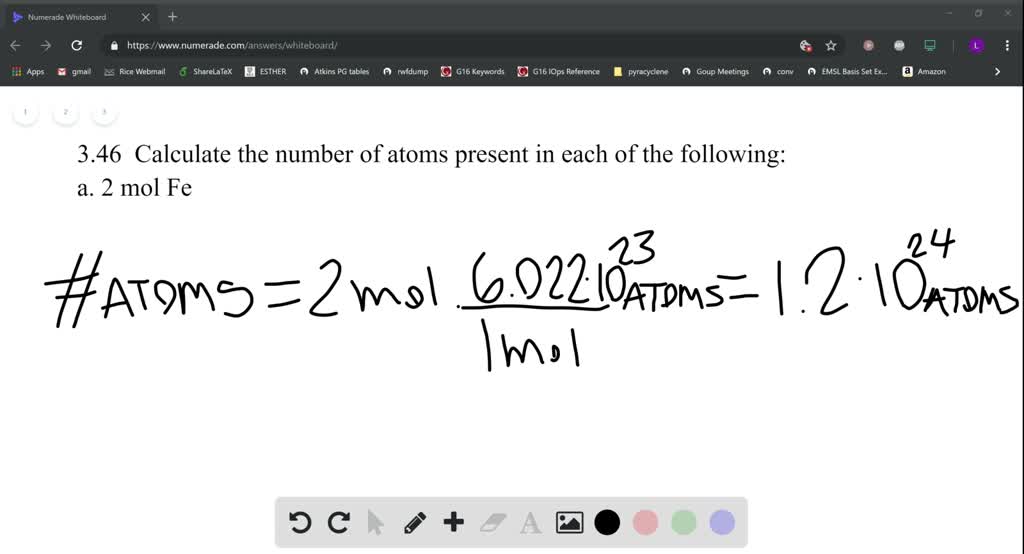
SOLVED:Calculate the number of atoms present in each of the following: a. 2 mol Fe b. 40.1 g Ca, which has an atomic mass of 40.08 amu c. 4.5 mol of boron-11

Calculate the number of atoms in each of the following:(i) 52 moles of Ar (ii) 52 u of He (iii) 52 g of He