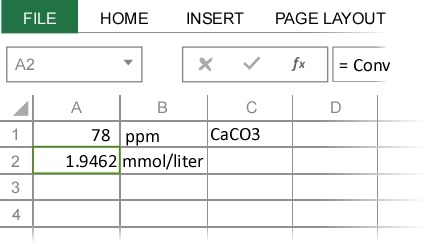
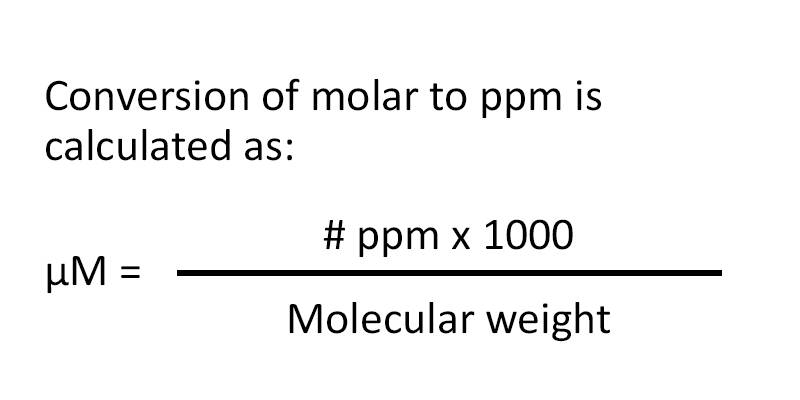
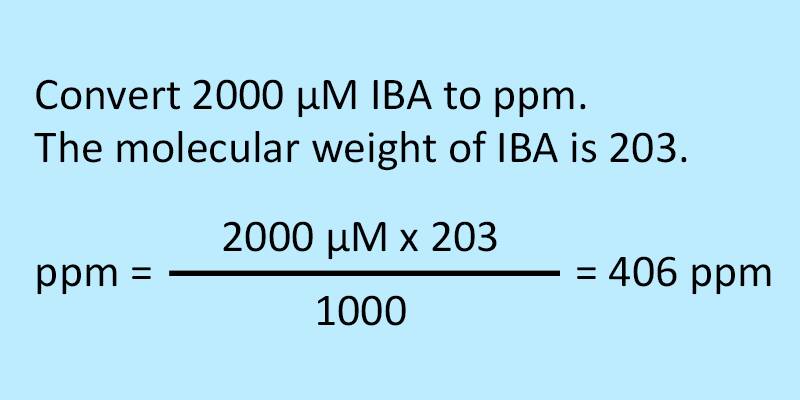
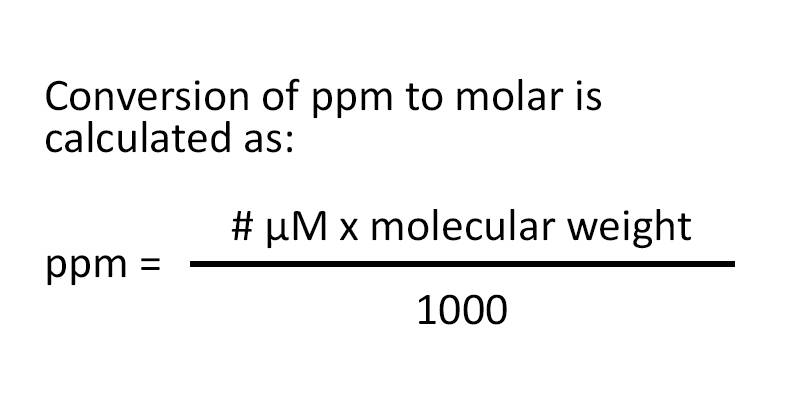
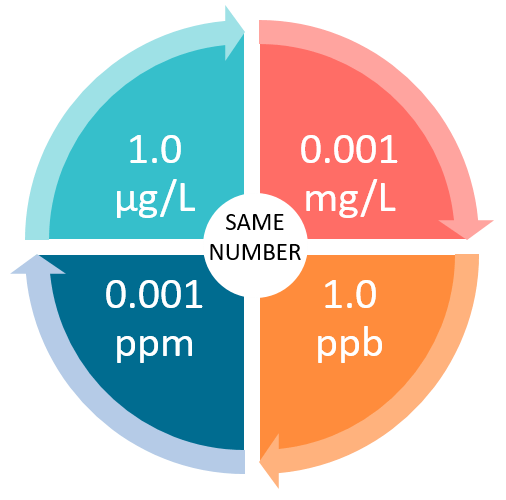SOLVED: Calculating the Water Hardness For convenience assume that all hardness is due to the presence of only Caz+ions in the form of calcium carbonate, CaCO3 The unit of hardness may be

1 Example A 25.0 L sample was found to contain 26.7 g glucose. Express the concentration as ppm and mg/dL glucose. Solution A ppm is defined as g/mL, - ppt download
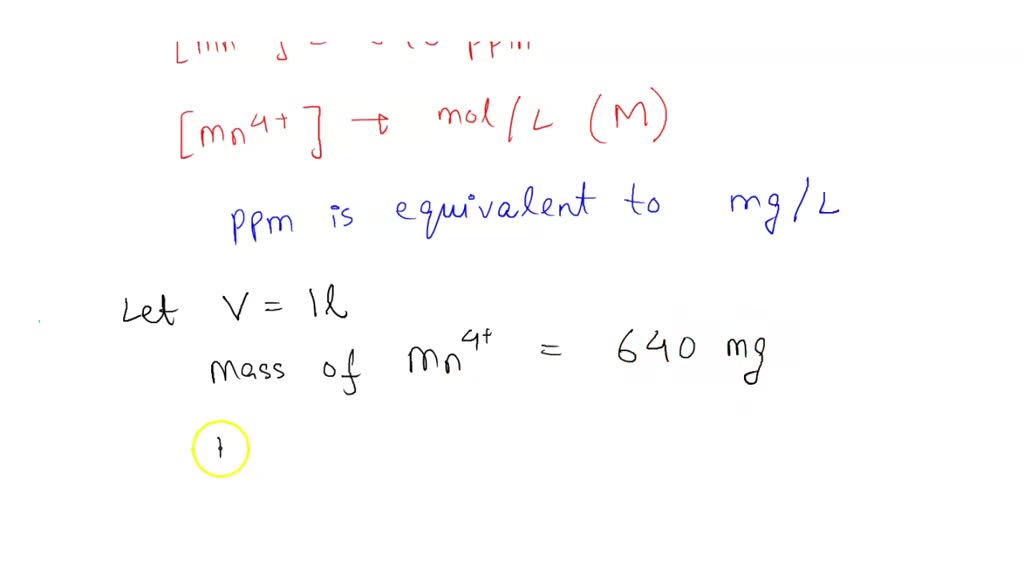
SOLVED: A contaminated sample of water contains 640 ppm of manganese ions, Mn4+ Calculate the concentration of manganese ions in mol/L. Show all work (/2)