6 versus 12 months of adjuvant trastuzumab for HER2-positive early breast cancer (PERSEPHONE): 4-year disease-free survival results of a randomised phase 3 non-inferiority trial - The Lancet

PDF) Immune modulation of pathologic complete response after neoadjuvant HER2-directed therapies in the NeoSphere trial

Neo Sphere - Formation professionnelle, 60 av Joseph Raynaud, 83140 Six Fours les Plages (France) - Adresse, Horaire

Incidence and management of diarrhea in patients with HER2-positive breast cancer treated with pertuzumab - Annals of Oncology

Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer ( NeoSphere) a randomised multicentre, open-label, phase 2 trial – OncoPrescribe

Immune modulation of pathologic complete response after neoadjuvant HER2-directed therapies in the NeoSphere trial - ScienceDirect

Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer ( NeoSphere): a randomised multicentre, open-label, phase 2 trial - The Lancet Oncology

YuGiOh Trading Card Game Turbo Pack Booster Five Single Card Common Darkness Neosphere TU05-EN016 - ToyWiz
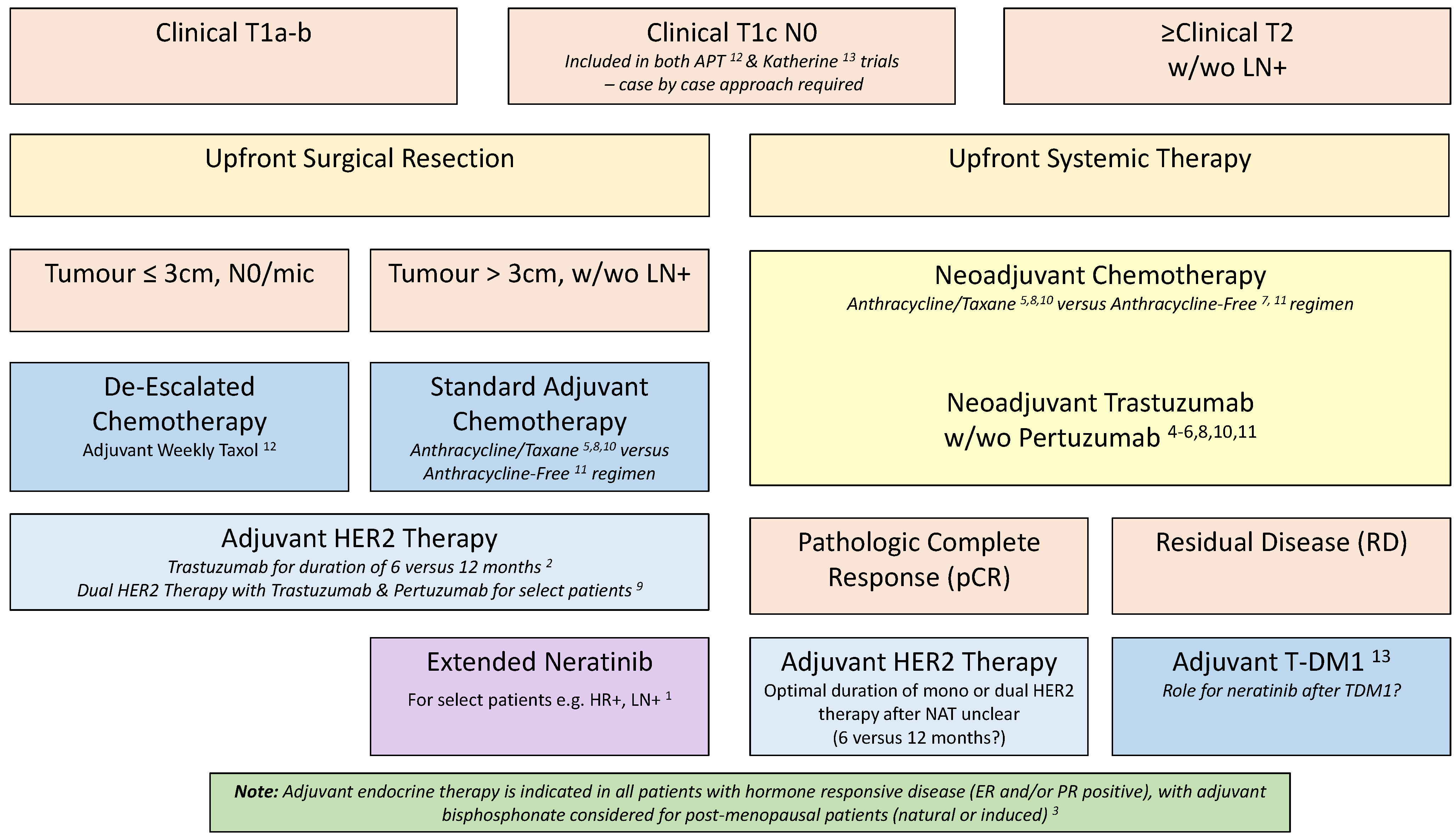
Current Oncology | Free Full-Text | Evolving Role of Risk Tailored Therapy in Early Stage HER2-Positive Breast Cancer: A Canadian Perspective
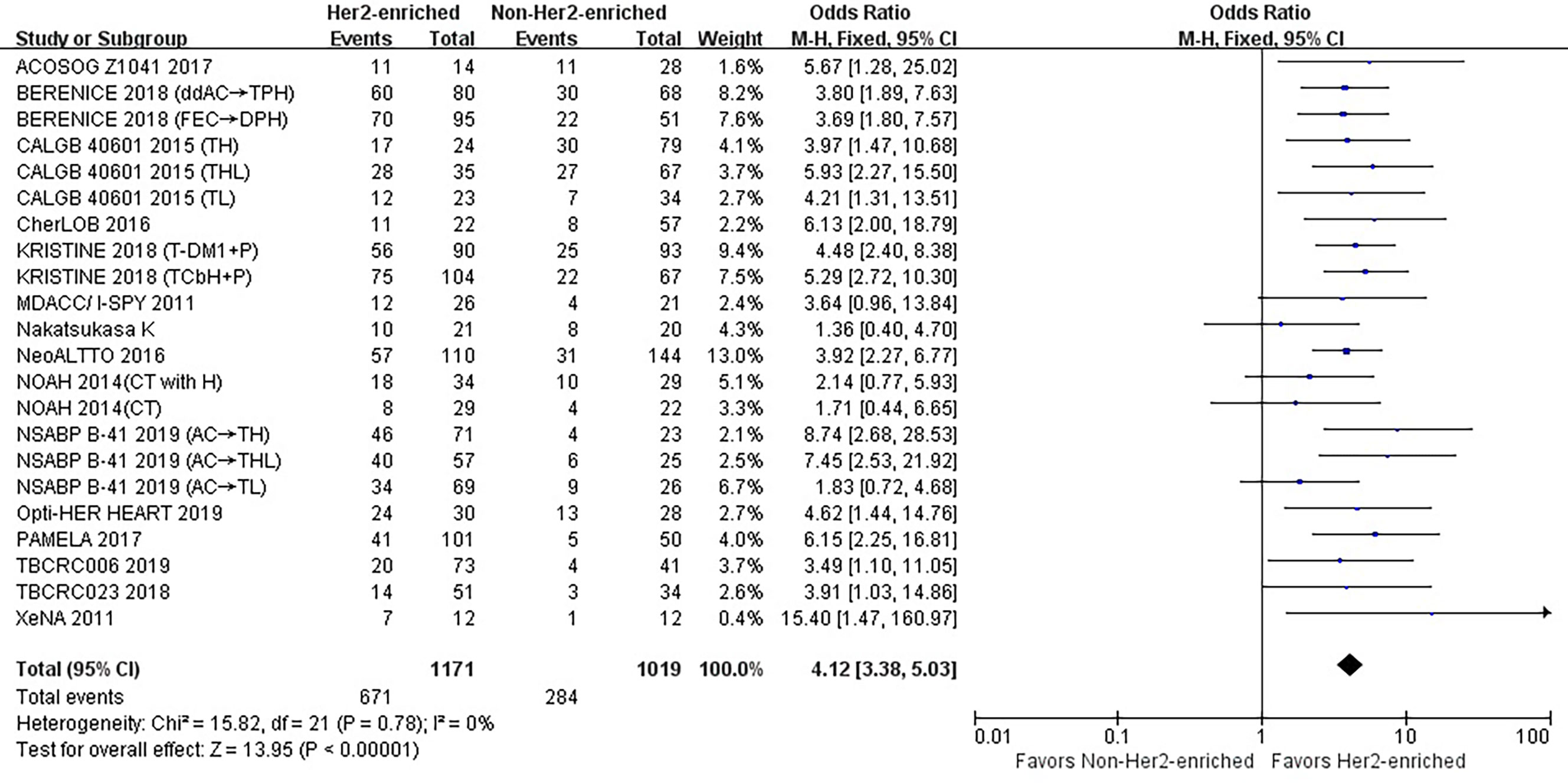
Frontiers | Meta-Analysis of HER2-Enriched Subtype Predicting the Pathological Complete Response Within HER2-Positive Breast Cancer in Patients Who Received Neoadjuvant Treatment