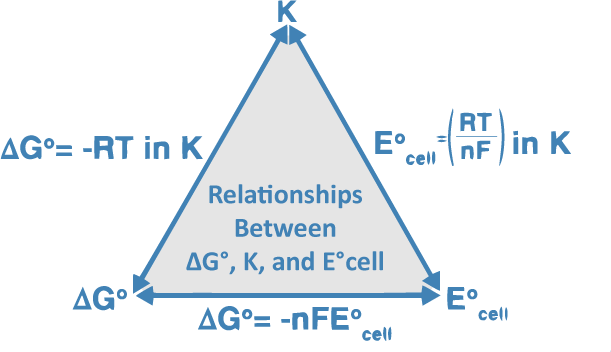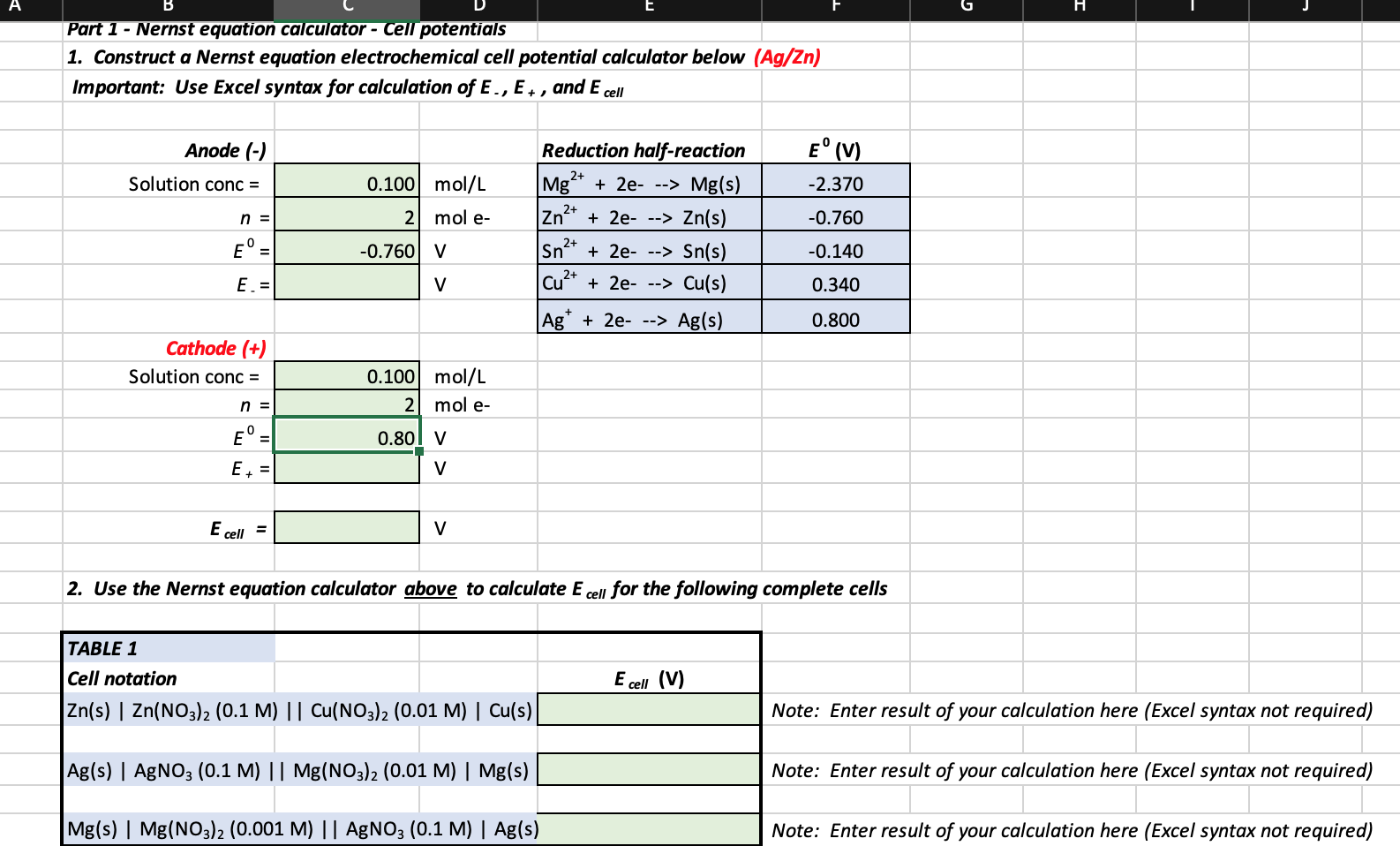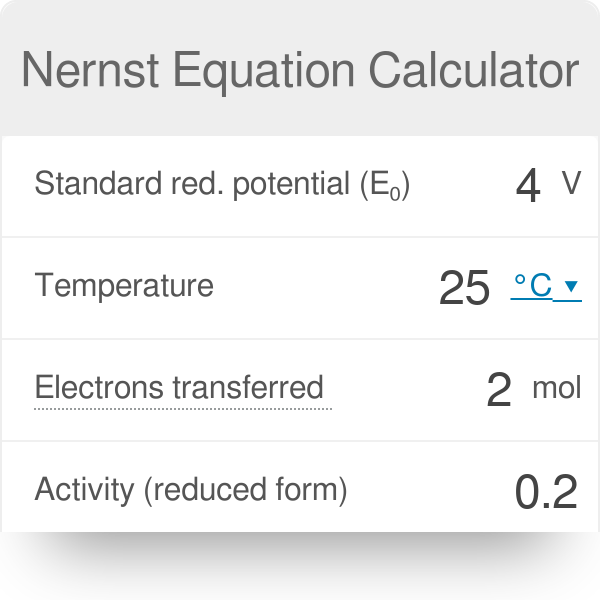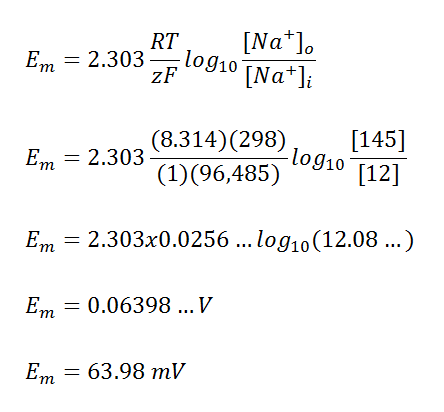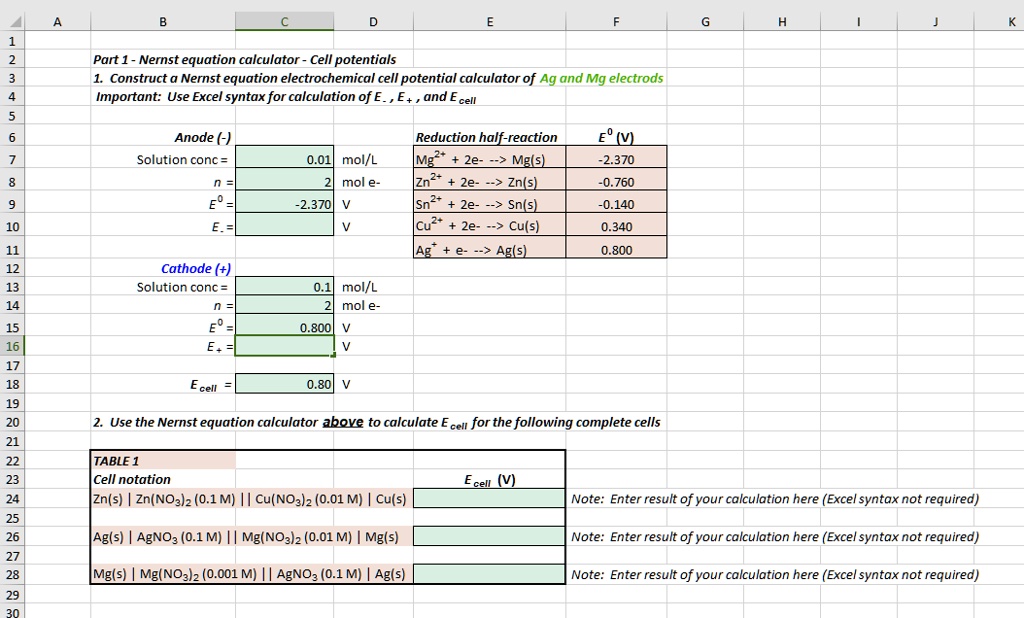
SOLVED: Part 1 Nernst equation calculator Cell potentials Construct Nernst equation electrochemical cell potential calculator of Ag and Mg electrods Important: Use Excel syntax for calculation of E and Ecell Anode (-)
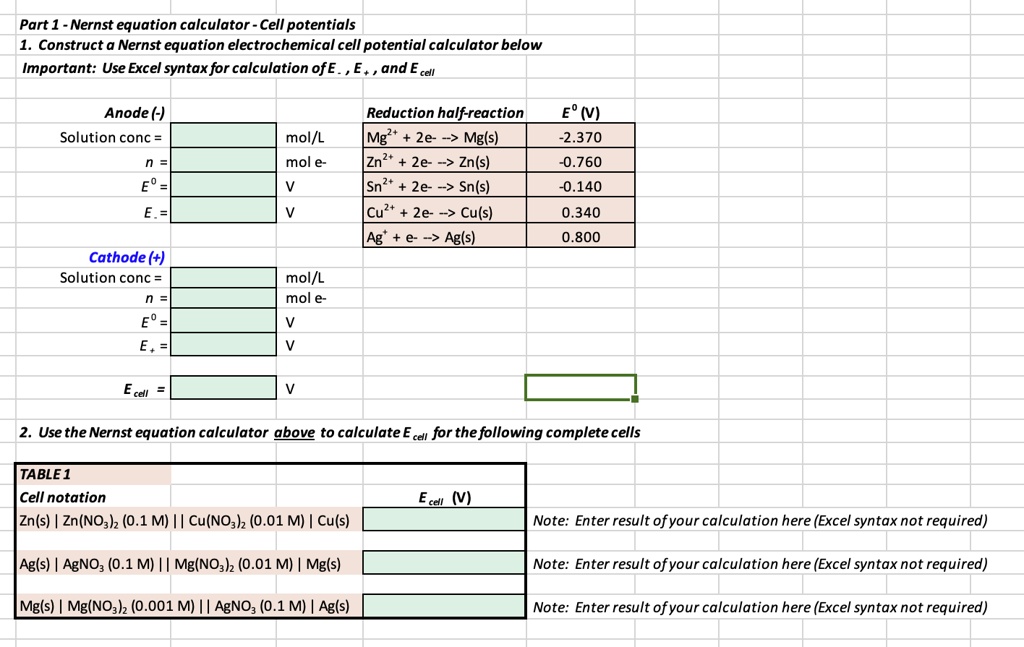
SOLVED: Part 1 Nernst equation calculator - Cell potentials Construct a Nernst equation electrochemical cell potential calculator below Important: Use Excel syntaxfor calculation ofE and E cel Anode (-) Solution conc Reduction

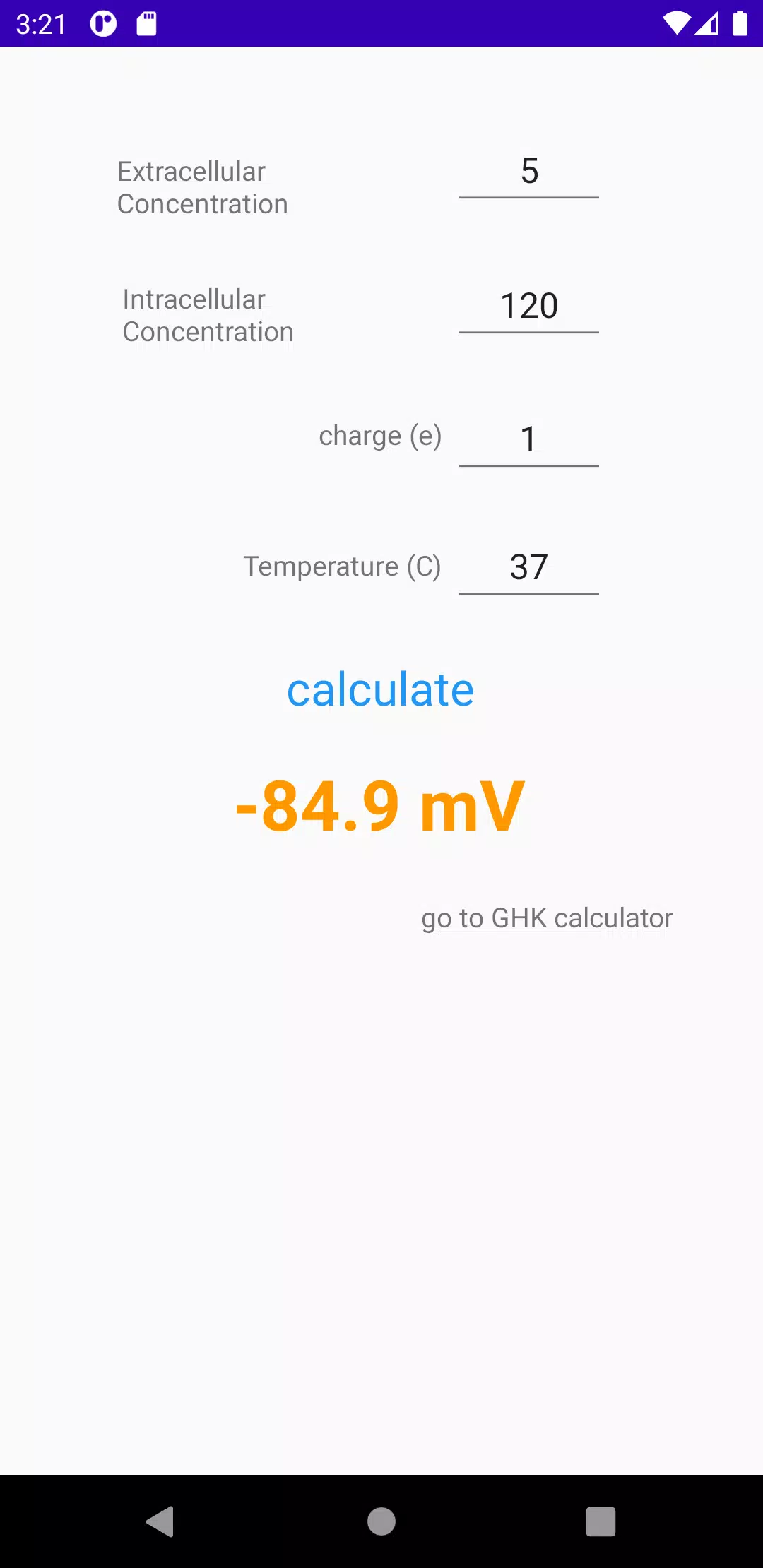
Numerical calculations: Nernst equilibrium potential calculator. The... | Download Scientific Diagram