Explain the variation of basic strength in: NH3 PH3 AsH3 SbH3 - Chemistry - Classification of Elements and Periodicity in Properties - 15225047 | Meritnation.com

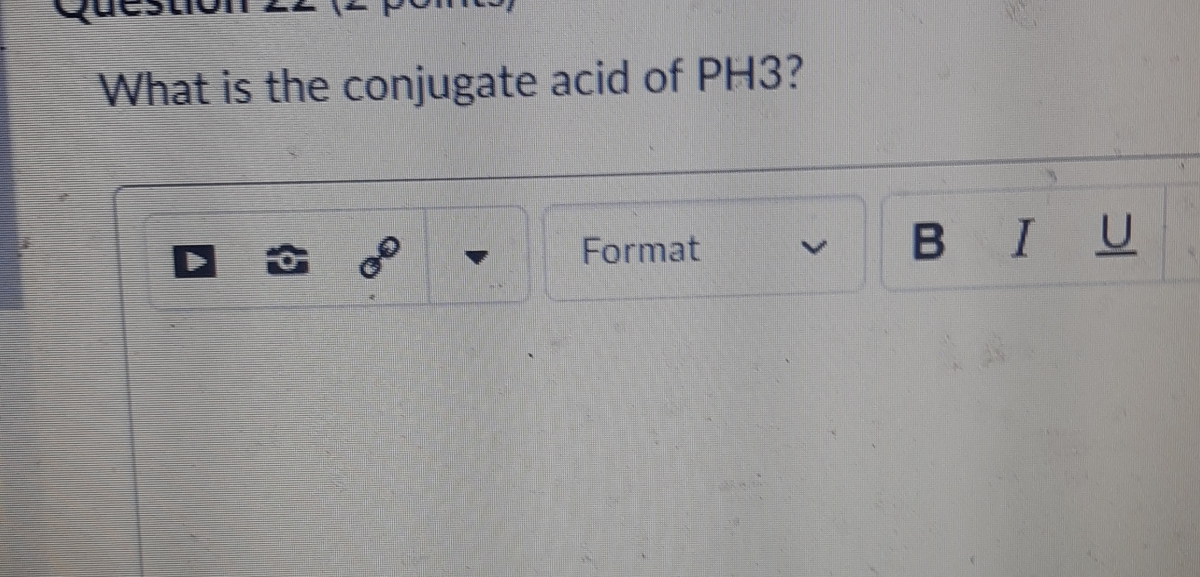
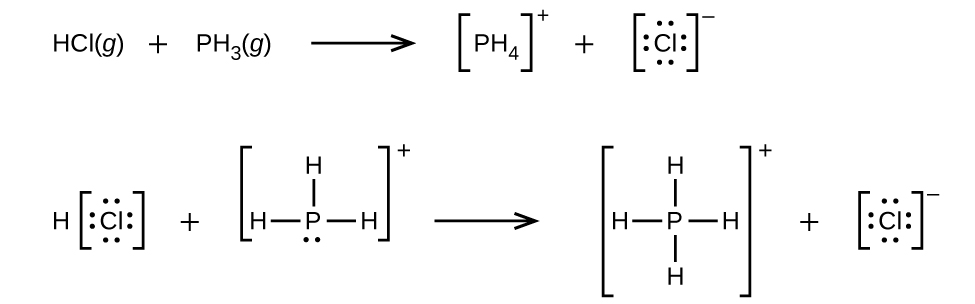
SOLVED: Identify the Lewis base in the following reaction: PH3(g) + H+(g) —> PH4+(g) Select one: a. None of these is a base b. PH4+ c. All of these are bases. d.

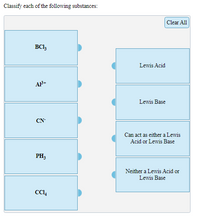
Which of the following statements does not support the fact that NH3 is a stronger Lewis base than PH3 ?
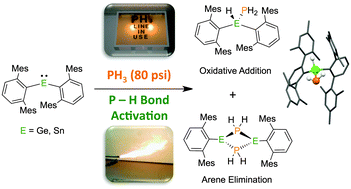
Activation of gaseous PH3 with low coordinate diaryltetrylene compounds - Chemical Communications (RSC Publishing)

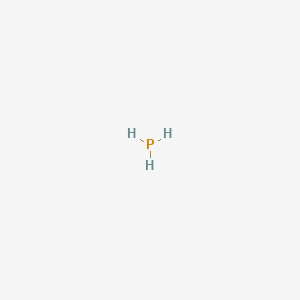
Draw the Lewis structure for PH3. How many bonds and non-bonding pairs are around the central atom, and what is the shape of this molecule? | Homework.Study.com

Draw the Lewis structure for PH3. How many bonds and non-bonding pairs are around the central atom, and what is the shape of this molecule? | Homework.Study.com