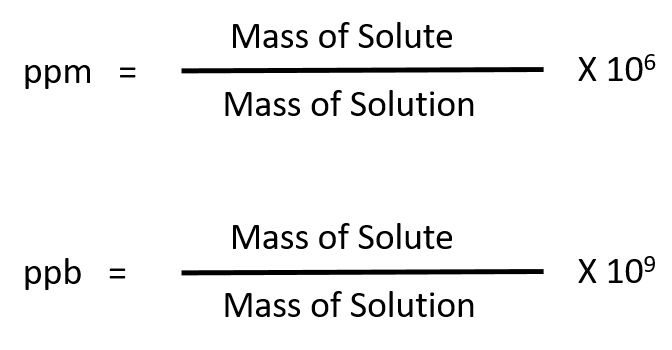
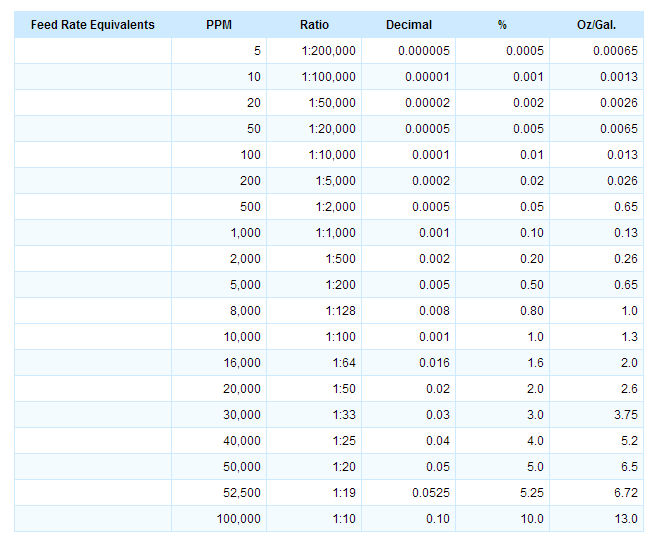
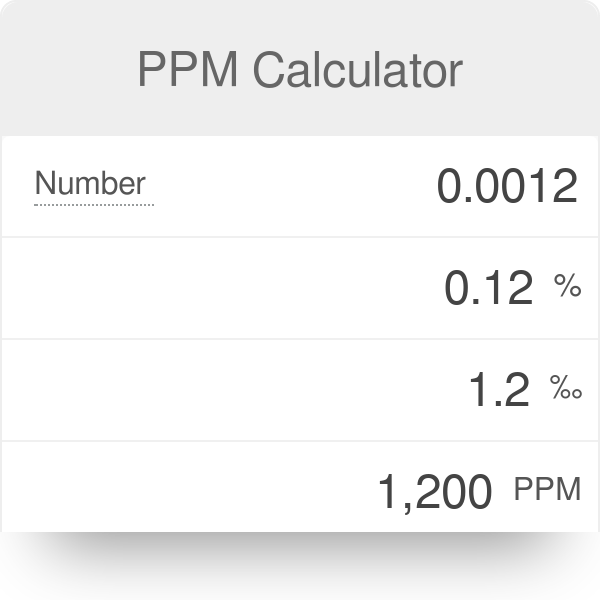
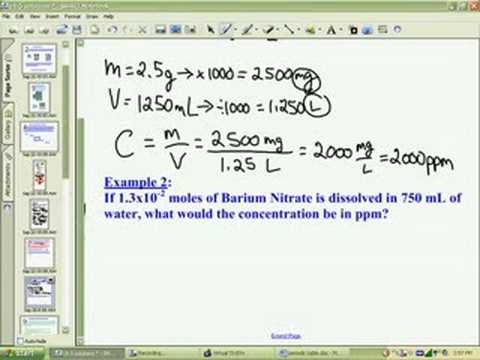
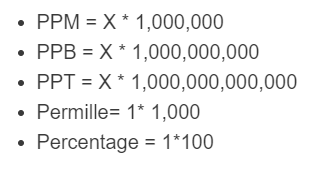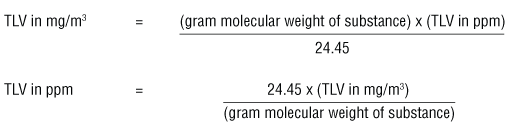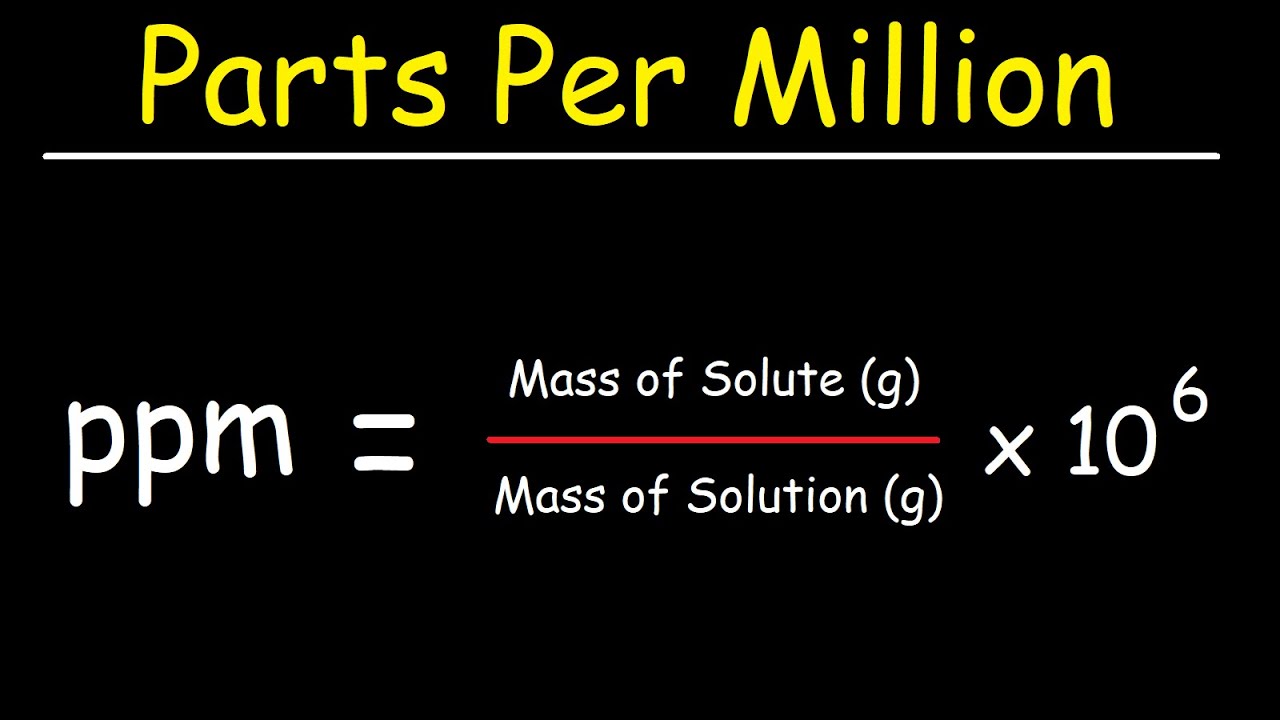![Grade 11 Chemistry, % Concentrations] Question 2- How do I calculate the amount of mg of 2.5 ppm in 5L of water? (Notes/formulas are shown on the second image but I don't Grade 11 Chemistry, % Concentrations] Question 2- How do I calculate the amount of mg of 2.5 ppm in 5L of water? (Notes/formulas are shown on the second image but I don't](https://preview.redd.it/ysaa5cvp7du51.jpg?width=3024&format=pjpg&auto=webp&s=5457a85ae64a9cf04410e9a99ac1681efcf7a493)
Grade 11 Chemistry, % Concentrations] Question 2- How do I calculate the amount of mg of 2.5 ppm in 5L of water? (Notes/formulas are shown on the second image but I don't

Calculate the concentration of nitric acid in moles per litre in a sample which has a density 1.41 g mL^-1 and the mass per cent of nitric acid in it being 69% .