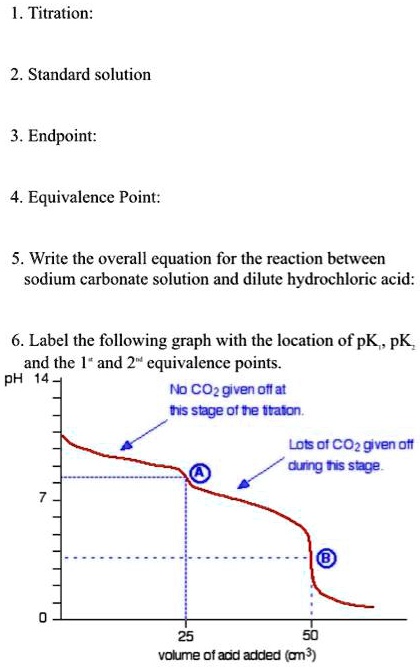
SOLVED: 1. Titration: Standard solution 3 . Endpoint: 4. Equivalence Point: 5. Write the overall equation for the reaction between sodium carbonate solution and dilute hydrochloric acid: 6. Label the 'following graph
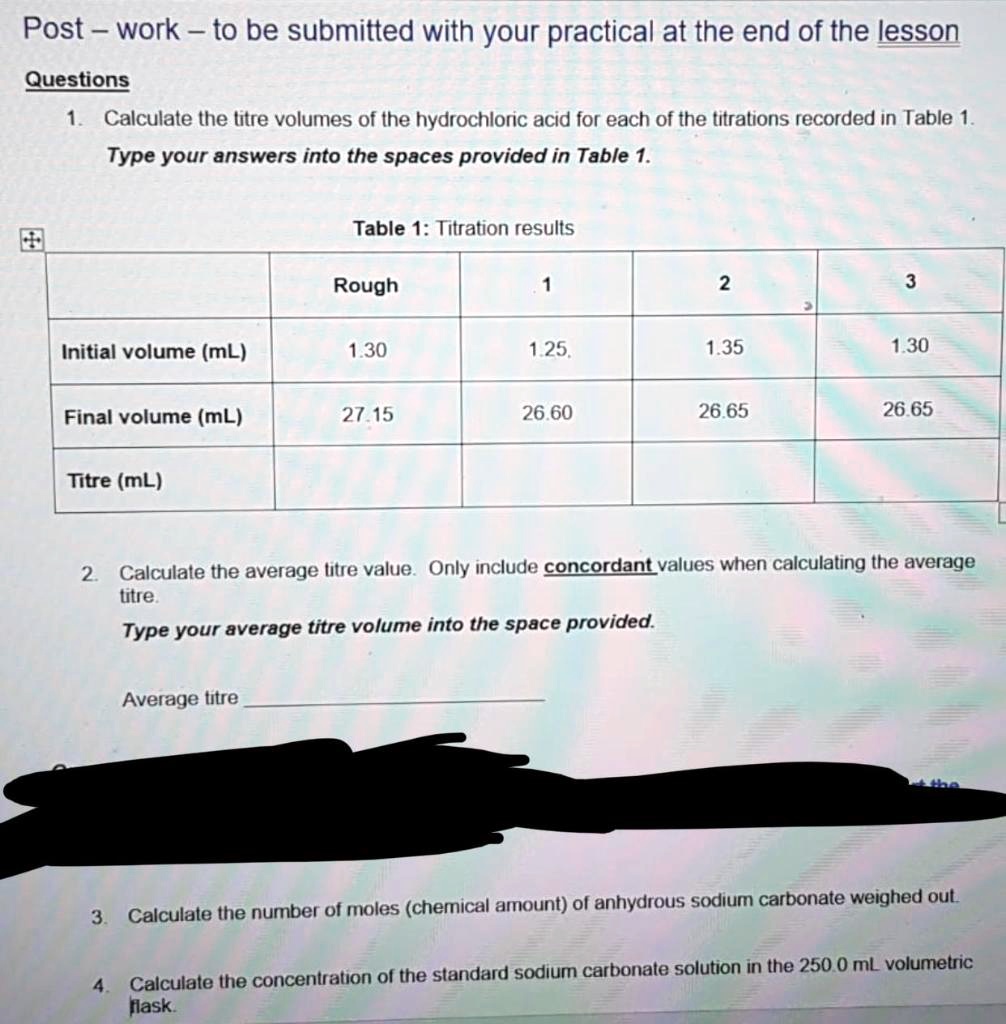
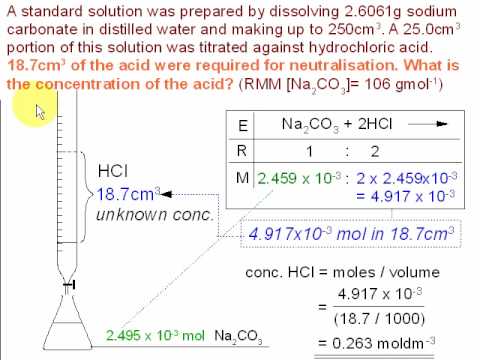
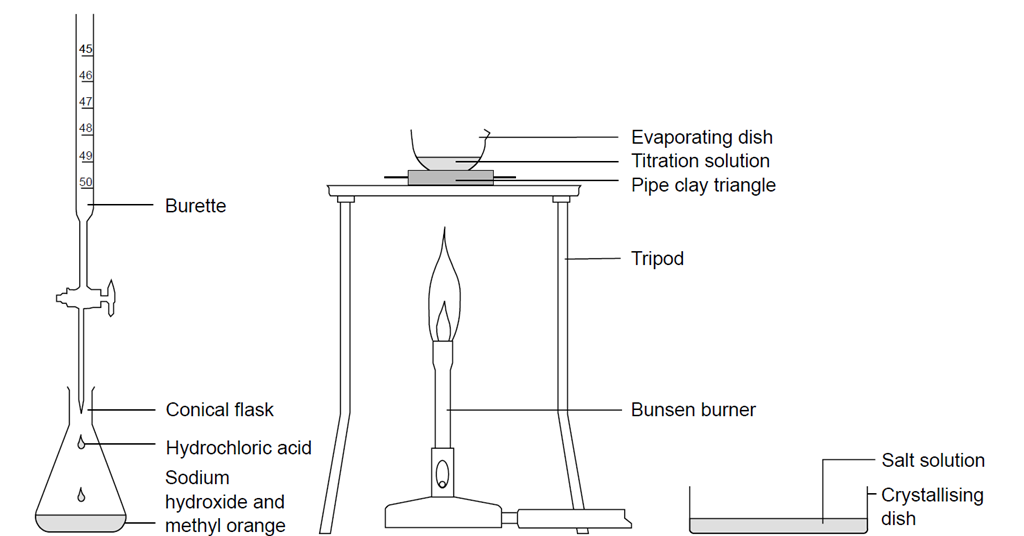
SOLVED: Post work to be submitted with your practical at the end of the lesson Questions Calculate the litre volumes of the hydrochloric acid for each of the titrations recorded in Table
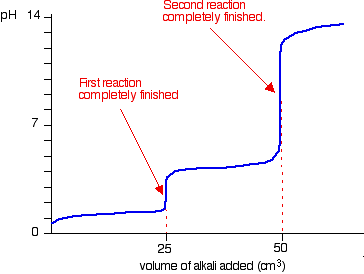
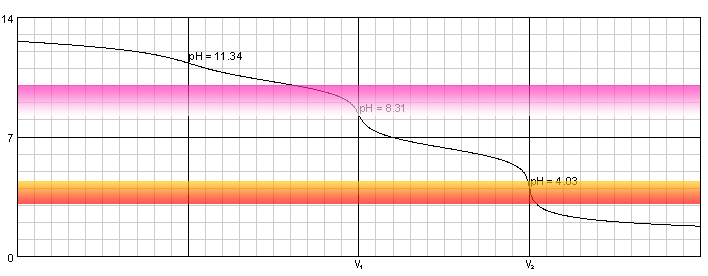
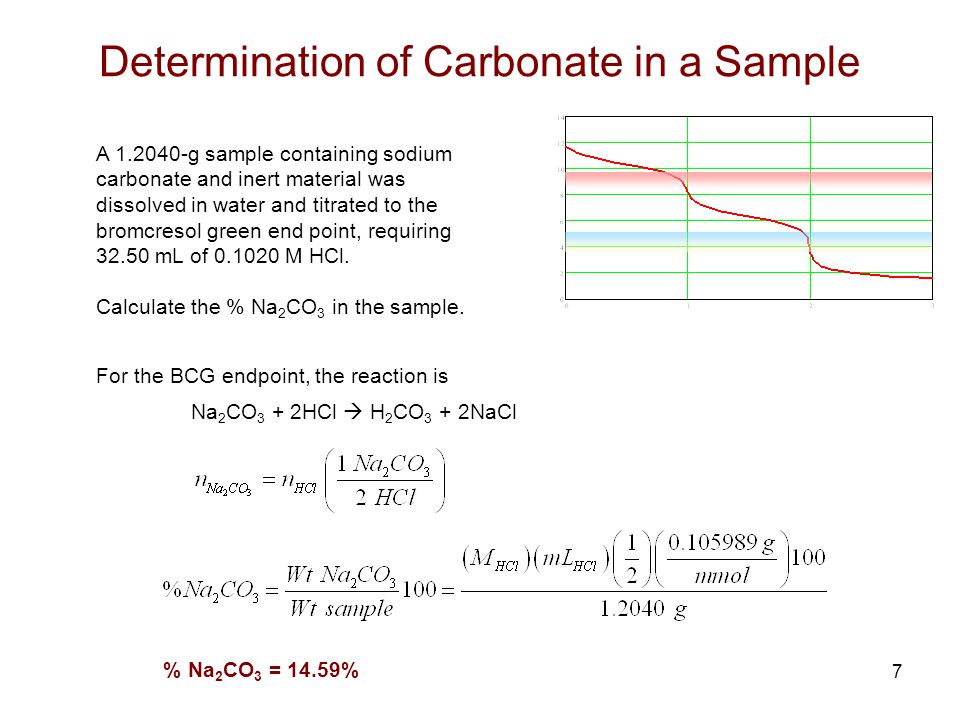
In the titration of hydrochloric acid with standard sodium carbonate solution, the 2nd endpoint should correspond twice the amount of the first. But I am not getting that kind of result. What's

Sodium Carbonate + Hydrochloric Acid - Na2CO3 + HCl - Molecular Equations & Net Ionic Equations - YouTube
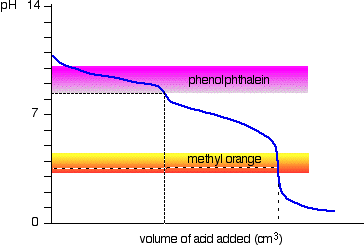
Explain how a pH meter could be used to find the exact volume of acid required to completely react with a sodium carbonate solution? - Quora

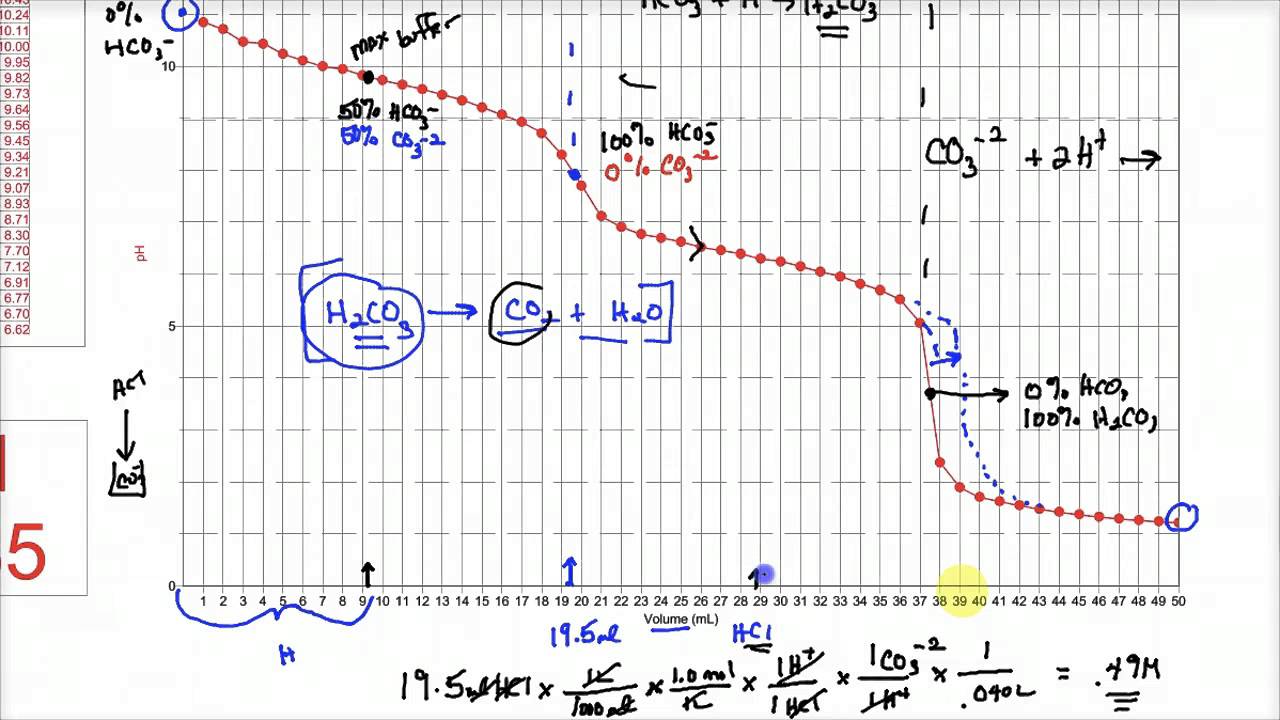
During the titration of sodium carbonate with H Cl, the dissolved carbonate ion will exist in three different forms; CO_3^{-2}, H CO_3^{-1}, and H_2 CO_3. During which part of the titration (initial,