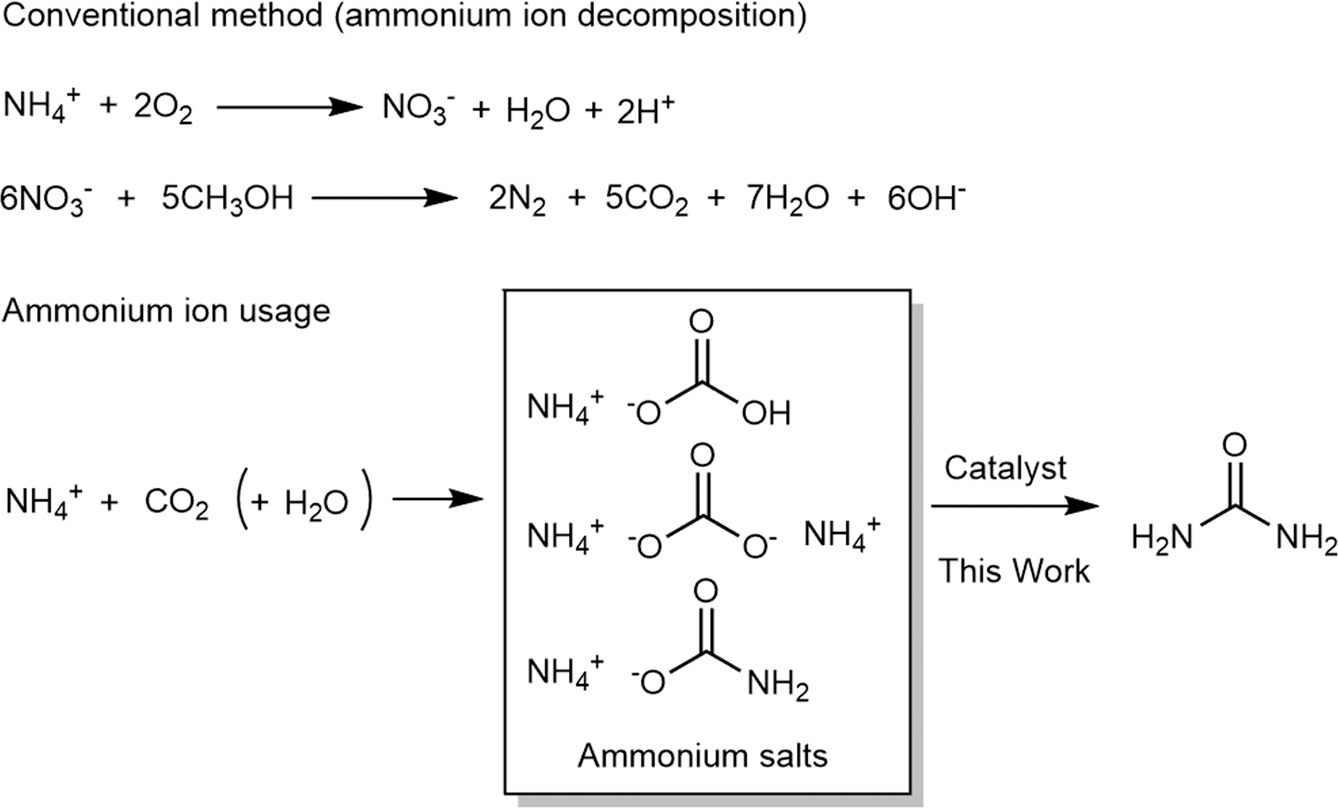
A novel method for molecular transformation to obtain energy from ammonia! | Nature Portfolio Chemistry Community

Question Video: Identifying the Lewis Acid in the Reaction of Ammonia with Boron Trifluoride | Nagwa

Highly efficient and selective separation of ammonia by deep eutectic solvents through cooperative acid-base and strong hydrogen-bond interaction - ScienceDirect